Key Points
Blood-specific expression of the Srsf2 P95H mutant results in decreased stem/progenitor cell numbers and a reduced repopulation capacity.
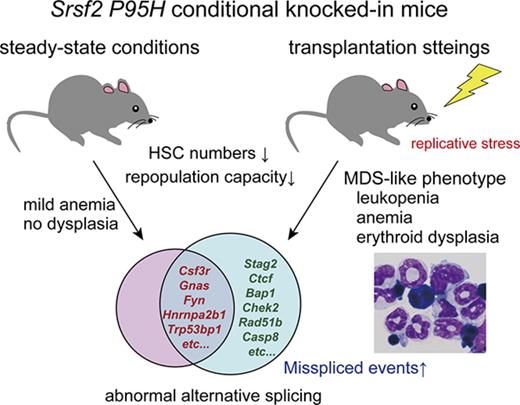
Srsf2 P95H mutation by itself is not sufficient to develop MDS but contributes to the MDS phenotype in transplantation settings.
Abstract
Splicing factor mutations are characteristic of myelodysplastic syndromes (MDS) and related myeloid neoplasms and implicated in their pathogenesis, but their roles in the development of MDS have not been fully elucidated. In the present study, we investigated the consequence of mutant Srsf2 expression using newly generated Vav1-Cre–mediated conditional knockin mice. Mice carrying a heterozygous Srsf2 P95H mutation showed significantly reduced numbers of hematopoietic stem and progenitor cells (HSPCs) and differentiation defects both in the steady-state condition and transplantation settings. Srsf2-mutated hematopoietic stem cells (HSCs) showed impaired long-term reconstitution compared with control mice in competitive repopulation assays. Although the Srsf2 mutant mice did not develop MDS under the steady-state condition, when their stem cells were transplanted into lethally irradiated mice, the recipients developed anemia, leukopenia, and erythroid dysplasia, which suggests the role of replicative stress in the development of an MDS-like phenotype in Srsf2-mutated mice. RNA sequencing of the Srsf2-mutated HSPCs revealed a number of abnormal splicing events and differentially expressed genes, including several potential targets implicated in the pathogenesis of hematopoietic malignancies, such as Csf3r, Fyn, Gnas, Nsd1, Hnrnpa2b1, and Trp53bp1. Among the mutant Srsf2-associated splicing events, most commonly observed were the enhanced inclusion and/or exclusion of cassette exons, which were caused by the altered consensus motifs for the recognition of exonic splicing enhancers. Our findings suggest that the mutant Srsf2 leads to a compromised HSC function by causing abnormal RNA splicing and expression, contributing to the deregulated hematopoiesis that recapitulates the MDS phenotypes, possibly as a result of additional genetic and/or environmental insults.
Introduction
Myelodysplastic syndromes (MDS) and related neoplasms, including chronic myelomonocytic leukemia (CMML), are stem cell–derived chronic myeloid neoplasms, characterized by abnormal blood cell morphologic status and ineffective hematopoiesis leading to blood cytopenias (myelodysplasia).1 Progression to secondary acute myeloid leukemia is common and is found in approximately one-third of patients.2 As for their pathogenesis, high-throughput genomic studies of recent years have revealed frequent pathway mutations involving multiple components of the RNA splicing machinery in myelodysplasia, which have been shown to be among the most frequently mutated classes of genes in these neoplasms.3,4 Largely occurring in a mutually exclusive manner, most of these mutations affect the components that are involved in the initial steps of premessenger RNA splicing, including SF3B1, SRSF2, U2AF1, and ZRSR2, which suggest their common effect on RNA splicing in the pathogenesis of myelodysplasia.5
Among frequently mutated splicing factors (SFs), SRSF2 is a member of the serine/arginine (SR) family of proteins that is characterized by 1 or 2 RNA recognition motifs (RRMs) and a serine/arginine-rich (RS) domain. SRSF2 binds RNA motifs known as exonic splicing enhancers within the target exon through an RRM and promotes inclusion or exclusion of that exon, playing an indispensable role in both constitutive and alternative splicing (AS) in most cell types.6-8 Somatic mutations in SRSF2 occur most frequently in CMML (28%-47%),3-5,9,10 where SRSF2 mutations represent one of the initiating events during MDS pathogenesis.3,4,11 All SRSF2 mutations are heterozygous and almost always affect the proline 95 residue within an intervening sequence between the RRMs and the RS domain, which suggests a neomorphic function of these mutations.
Recently, several studies have attempted to clarify the role of SF mutations in vivo. Mx1-Cre–mediated conditional knockin of the Srsf2 P95H mutation impaired hematopoietic differentiation, increased hematopoietic stem and progenitor cells (HSPCs), and promoted myelodysplasia by altering the RNA-binding specificity of Srsf2 in mice.12 The altered RNA-binding affinity of mutant SRSF2 and splicing changes were also demonstrated using leukemic cell lines expressing an SRSF2 mutant allele.13 Other groups have reported that mice models of the pathogenic mutations in U2af114 and Sf3b115,16 displayed defects in differentiation accompanied by RNA-splicing changes. These studies represent major advances toward understanding how SF mutations affect hematopoietic functions in myelodysplasia. However, given that the hematological phenotypes were investigated only in transplantation settings in the published Srsf2 knockin mice model,12 it is unclear to what extent the observed phenotypes were ascribed to the effect of transplantation, which are known to cause considerable stress on hematopoietic cells and an altered bone marrow (BM) microenvironment. Thus, in this study, we investigated the hematological consequences of Srsf2 mutation both under the steady-state condition and in a regenerative context using a newly generated Vav1-Cre–mediated conditional knockin mouse model of the Srsf2 P95H mutation.
Materials and methods
Mice
We constructed the Srsf2-targeting vector using the “flip-excision” (FLEx) switch strategy as previously described.17,18 The linearized vector was electroporated into murine hybrid (C57BL/6×129/Sv) embryonic stem cells. Chimeric mice were produced by microinjection of targeted embryonic stem cells into blastocysts and were bred to C57BL/6 mice to establish germ line transmission. Mice were backcrossed against C57BL/6 mice for at least 6 generations. All animal experiments were approved by the Animal Care and Use Committee of the Institute of Medical Science, the University of Tokyo, and Kyoto University. Full details are provided in supplemental Methods.
Flow cytometry and BM transplantation
Single-cell suspensions prepared from the BM, spleen, and peripheral blood (PB) were stained with monoclonal antibodies (supplemental Table 1). Purification of HSPCs was performed as previously described.19 In competitive or noncompetitive BM transplantation, 1 × 106 freshly isolated BM cells were transplanted into lethally irradiated recipient mice with or without the same number of competitor BM cells. Full details are described in supplemental Methods.
RNA sequencing
We extracted total RNA using NucleoSpin RNA XS (Macherey-Nagel) from sorted cell populations from 17- to 21-week-old littermate Srsf2 mutant and wild-type mice (steady-state) or recipient mice 3 to 6 months after noncompetitive BM transplantation. RNA samples with RNA integrity number >8 proceeded to the sequencing analysis. The synthesis and amplification of complementary DNA (cDNA) were performed using SMARTer Ultra Low Input RNA Kit for Sequencing, version 3 or 4 (Clontech). Sequencing libraries were generated using the Low Input Library Prep Kit (Clontech), followed by high-throughput sequencing on the HiSeq 2500 System (Illumina) with 124 bp paired-end reads.
For the data analysis, the sequencing reads were aligned to the mouse reference genome (mm10) using HISAT2 (version 2.0.4).20 To identify differential AS events, we applied rMATS21 with the following parameters: anchor length 2, unpaired analysis type, and “unstranded“ library type. Gene and transcript annotations were referred from “archive-2015-07-17-14-33-2“ in the University of California Santa Cruz annotation archives. Splicing site sequences and their logos were analyzed with Bioconductor seqLogo library (version 3.3). For differential expression analysis, transcript read counts mapped to each gene were extracted using Rsubread package22 and were compared using edgeR package.23,24 RNA sequencing (RNA-seq) data have been deposited in the DNA Data Bank of Japan repository under accession number DRA006224.
Statistical analysis
We calculated P values comparing 2 means using the 2-tailed unpaired Student t test using GraphPad Prism, version 6. The log-rank test was used to compare the overall survival. We used the Fisher’s exact test to determine the P values comparing the composition of SSNG motifs in the cassette exons (CEs). Statistical significance of the overlap between differentially spliced or expressed genes from different cell populations was estimated using Monte Carlo simulations.
Results
Reduced number of HSPCs with impaired differentiation in Srsf2 mutant mice
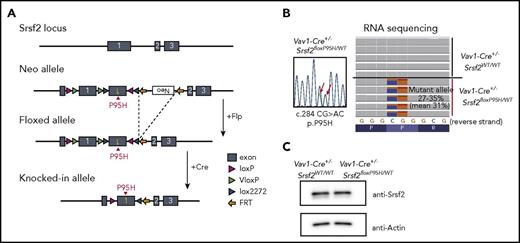
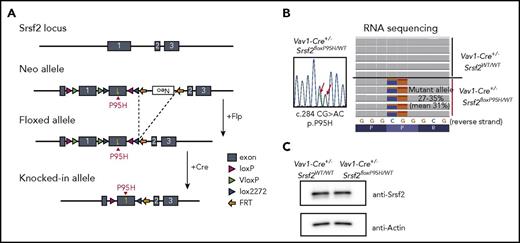
To elucidate the role of SRSF2 mutation in the development of myelodysplasia, we generated an Srsf2P95H conditional knock-in mouse using a FLEx switch strategy (Figure 1A).18 We chose Vav1-Cre transgenic mice, instead of Mx1-Cre mice, as a Cre deleter strain to induce constitutive and hematopoietic-specific Cre expression25 to exclude the proproliferative effects of interferon.26,27 Mice heterozygous for the floxed Srsf2P95H allele were crossed with Vav1-Cre mice to obtain a cohort of Vav1-Cre+/−Srsf2floxP95H/WT and control Vav1-Cre+/−Srsf2WT/WT mice. Vav1-Cre+/−Srsf2floxP95H/WT mice expressed nearly equal levels of Srsf2P95H (c.284CG>AC) and wild-type Srsf2 alleles in hematopoietic cells, resulting in equivalent levels of Srsf2 protein expression compared with Vav1-Cre+/−Srsf2WT/WT mice (Figure 1B-C). Successful recombination and expression of the Srsf2P95H allele were also confirmed by genotyping of multiple colonies (14/14) derived from single CD34−KSL HSCs. Notably, Vav1-Cre–positive mice having homozygous Srsf2floxP95H alleles were not obtained (0/43), which suggests the lethality of homozygous expression of the mutant Srsf2 allele. Unexpectedly, however, we also failed to obtain the mice having homozygous floxed Srsf2P95H alleles, even under the Vav1-Cre–negative background (0/46), which suggests that the floxed Srsf2P95H allele was null or severely hypomorphic due to the FLEx switch design. In fact, Vav1-Cre–negative Srsf2floxP95H/WT mice were born normally, but RNA-seq of their BM cells revealed a low level of Srsf2 expression from the floxed allele (∼2.0% of that from the wild-type allele) (supplemental Figure 1A), although the mice had an apparently normal level of wild-type Srsf2 expression with a trace amount of the mutant Srsf2 reads (∼1.1%), probably caused by spontaneous recombination. Thus, it was difficult to determine whether the lethality of Vav1-Cre–positive Srsf2floxP95H/floxP95H mice was due to homozygous expression of the mutant Srsf2 allele in hematopoietic tissues or to the Srsf2 deficiency in other tissues with low Vav1 promoter activities, or to both.
Generation of Srsf2 P95H conditional knockin mice. (A) Schematic representation of the targeted Srsf2 allele. (B) Sanger sequencing of polymerase chain reaction products of cDNA of the BM cells from Vav1-Cre+/−Srsf2floxP95H/WT mice (left), and RNA-seq of KSL cells of Vav1-Cre+/−Srsf2WT/WT and Vav1-Cre+/−Srsf2floxP95H/WT mice (right). (C) Western blot analysis of the expression of Srsf2 in the BM-derived nucleated cells of Vav1-Cre+/−Srsf2WT/WT and Vav1-Cre+/−Srsf2floxP95H/WT mice. FRT, flippase recognition target.
Generation of Srsf2 P95H conditional knockin mice. (A) Schematic representation of the targeted Srsf2 allele. (B) Sanger sequencing of polymerase chain reaction products of cDNA of the BM cells from Vav1-Cre+/−Srsf2floxP95H/WT mice (left), and RNA-seq of KSL cells of Vav1-Cre+/−Srsf2WT/WT and Vav1-Cre+/−Srsf2floxP95H/WT mice (right). (C) Western blot analysis of the expression of Srsf2 in the BM-derived nucleated cells of Vav1-Cre+/−Srsf2WT/WT and Vav1-Cre+/−Srsf2floxP95H/WT mice. FRT, flippase recognition target.
None of the Vav1-Cre+/−Srsf2floxP95H/WT mice developed MDS or other hematological malignancies at least by 90 weeks of age (0/20). There was no significant difference in overall survival between mutant and control mice (supplemental Figure 1B). Vav1-Cre+/−Srsf2floxP95H/WT mice exhibited decreased hemoglobin levels with elevated levels of mean corpuscular volume (MCV) and mean corpuscular hemoglobin (MCH) compared with Vav1-Cre+/−Srsf2WT/WT mice but otherwise showed grossly normal steady-state hematopoiesis (Figure 2A; supplemental Figure 1C). BM and spleen cellularity were similar between both mice, without showing morphologic abnormalities (Figure 2B; supplemental Figure 1D-F).
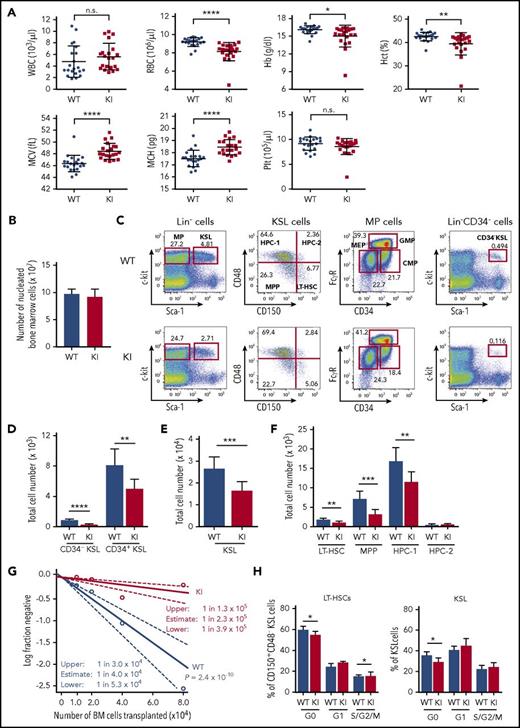
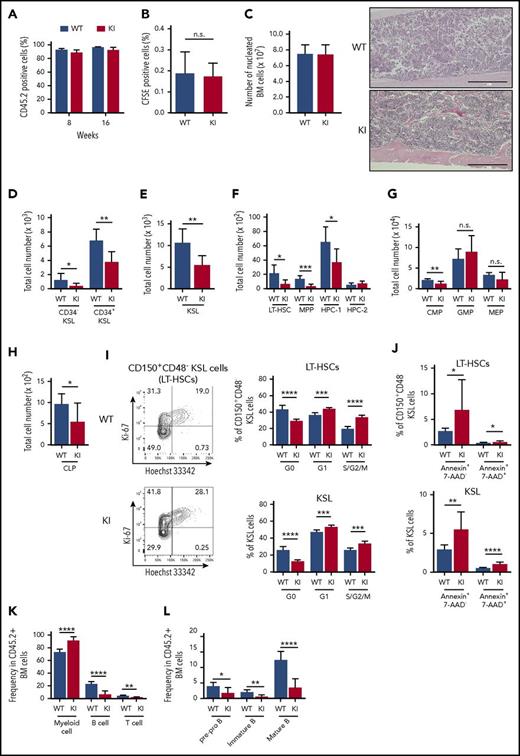
Reduced number of HSPCs with impaired differentiation in Srsf2 mutant mice. (A) White blood cells (WBCs), red blood cells (RBCs), hemoglobin (Hb), hematocrit (Hct), MCV, MCH, and platelet (Plt) counts in PB from 8- to 15-week-old Vav1-Cre+/−Srsf2WT/WT (WT) and Vav1-Cre+/−Srsf2floxP95H/WT (KI) littermate male mice are plotted as dots (n = 21 per group). The mean ± standard deviation (SD) are indicated as bars. (B) Absolute numbers of nucleated BM cells in bilateral femurs and tibias from 12- to 15-week-old WT and KI male mice are shown as mean ± SD (n = 8). (C) Representative flow cytometry analysis of the BM stem and progenitor populations of WT and KI mice. (D-F) Total cell number of CD34−KSLs and CD34+KSLs (panel D); KSL cells (panel E); and LT-HSCs, MPPs, HPC-1, and HPC-2 (panel F) in the BM of 12- to 15-week-old mice (mean ± SD, n = 8). (G) Limiting-dilution analysis based on competitive repopulation 3 months after transplantation determined the frequency of multilineage repopulating cells in the BM. Data are expressed as mean and 95% confidence intervals for WT (n = 96 samples) and KI (n = 95 samples) combined from 4 independent experiments. Frequency and probability estimates were computed using the extreme limiting dilution analysis (ELDA) software (Bioinformatics). (H) Cell cycle analysis in LT-HSCs (c-Kit+Sca-1+LinlowCD150+CD48−) and KSL cells using Ki-67 and Hoechst 33342 staining (mean ± SD, n = 6). *P < .05; **P < .01; ***P < .001; ****P < .0001. n.s., not significant.
Reduced number of HSPCs with impaired differentiation in Srsf2 mutant mice. (A) White blood cells (WBCs), red blood cells (RBCs), hemoglobin (Hb), hematocrit (Hct), MCV, MCH, and platelet (Plt) counts in PB from 8- to 15-week-old Vav1-Cre+/−Srsf2WT/WT (WT) and Vav1-Cre+/−Srsf2floxP95H/WT (KI) littermate male mice are plotted as dots (n = 21 per group). The mean ± standard deviation (SD) are indicated as bars. (B) Absolute numbers of nucleated BM cells in bilateral femurs and tibias from 12- to 15-week-old WT and KI male mice are shown as mean ± SD (n = 8). (C) Representative flow cytometry analysis of the BM stem and progenitor populations of WT and KI mice. (D-F) Total cell number of CD34−KSLs and CD34+KSLs (panel D); KSL cells (panel E); and LT-HSCs, MPPs, HPC-1, and HPC-2 (panel F) in the BM of 12- to 15-week-old mice (mean ± SD, n = 8). (G) Limiting-dilution analysis based on competitive repopulation 3 months after transplantation determined the frequency of multilineage repopulating cells in the BM. Data are expressed as mean and 95% confidence intervals for WT (n = 96 samples) and KI (n = 95 samples) combined from 4 independent experiments. Frequency and probability estimates were computed using the extreme limiting dilution analysis (ELDA) software (Bioinformatics). (H) Cell cycle analysis in LT-HSCs (c-Kit+Sca-1+LinlowCD150+CD48−) and KSL cells using Ki-67 and Hoechst 33342 staining (mean ± SD, n = 6). *P < .05; **P < .01; ***P < .001; ****P < .0001. n.s., not significant.
However, the absolute number of multipotent self-renewing HSCs characterized by low CD34 expression (CD34−KSL cells) or c-Kit+Sca-1+LinlowCD150+CD48−phenotype (long-term HSCs [LT-HSCs]) was significantly reduced in mutant mice compared with that of the control mice; the same trend was observed for immature hematopoietic progenitors, including KSL cells, CD34+c-Kit+Sca-1+Linlow (CD34+KSL) cells, c-Kit+Sca-1+LinlowCD150−CD48−cells (multipotent progenitors [MPPs]), and restricted hematopoietic progenitor cells (HPC-1; c-Kit+Sca-1+LinlowCD150−CD48+) (Figure 2C-F).28-30 The reduced number of HSCs in mutant mice was also demonstrated by limiting-dilution assays, in which the estimated number of multilineage repopulating cells of Srsf2 wild-type and mutant mice was 1 in 4.0 × 104 and 2.3 × 105 BM cells, respectively (Figure 2G). Whereas the frequency of apoptotic cells among LT-HSCs and KSL cells was not significantly changed (supplemental Figure 1G), the proportion of quiescent HSPCs was significantly decreased in Srsf2 mutant mice compared with control mice (Figure 2H), which indicates that the reduction in HSC numbers in mutant mice was due to the increase of the cycling HSCs. In contrast, there was no significant difference between mutant and control mice in the number of more differentiated progenitors, including common myeloid progenitors (CMPs; c-Kit+Sca-1−LinlowCD34+FcγRmed), granulocyte/macrophage lineage-restricted progenitors (GMPs; c-Kit+Sca-1−LinlowCD34+FcγRhigh), megakaryocyte/erythrocyte lineage-restricted progenitors (MEPs; c-Kit+Sca-1−LinlowCD34−FcγRlow),31 common lymphoid progenitors (CLPs; LinlowIL-7Rα+Flt3+Sca-1medc-Kitmed),32,33 and erythroid progenitors (supplemental Figure 1H-J). Lineage distribution in the BM and the spleen cells did not differ substantially between mutant and control animals either, except for impaired differentiation of B-cell precursors in mutant mice (supplemental Figure 1K-L).
Reduced number of HSPCs and dysplastic hematopoiesis in Srsf2 mutant transplanted mice
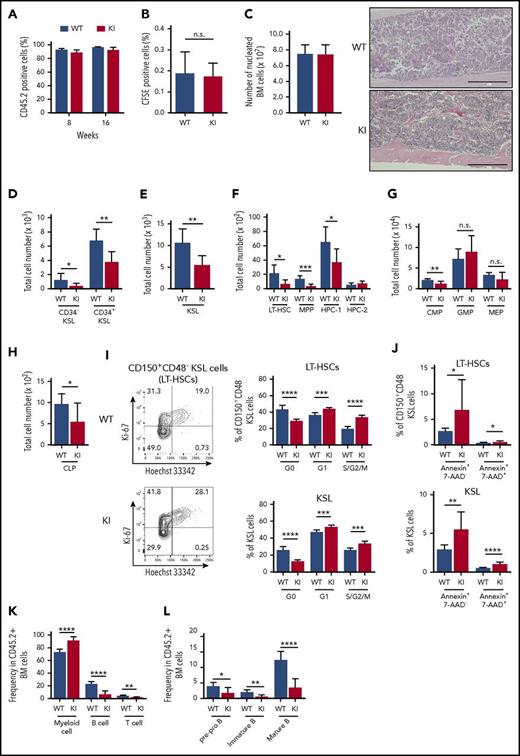
We next assessed the phenotype of Srsf2-mutated stem cells in transplantation settings to evaluate the effect of increasing replicative stress, which has been shown to substantially affect the behavior of normal and abnormal stem cells.34-36 Mutant-derived BM cells reconstituted the recipient’s hematopoiesis, in which more than 90% of PB cells were derived from donor cells at least until 16 weeks after transplantation (Figure 3A). In vivo homing assay using carboxyfluorescein succinimidyl ester (CFSE)–labeled BM-derived mononuclear cells showed no significant differences in the proportion of CFSE-positive cells in the recipient BM at 16 hours after transplantation, which suggests that mutant-derived BM cells largely retained normal homing capability (Figure 3B). There was no significant difference in BM cellularity between mutant and control transplanted mice, both displaying normocellular BM (Figure 3C). However, in agreement with the findings in the nontransplanted mice, flow cytometry of the BM from the mutant transplanted mice revealed a significant decrease in the numbers of stem and progenitor cells compared with the control transplanted mice. The reduced cell number was also observed in more mature hematopoietic fractions, including CMPs and CLPs (Figure 3D-H). The frequency of the cycling Srsf2 mutant HSCs was substantially higher than those of wild-type HSCs, and the difference became more prominent in transplantation settings compared with those in the steady-state condition (Figure 2H; Figure 3I). In addition, Srsf2 mutant HSCs showed a higher frequency of apoptosis, compared with wild-type HSCs (Figure 3J). The mutant transplanted mice showed an increase in the myeloid bias and impaired differentiation of B-cell precursors, as seen in human MDS (Figure 3K-L).37
Consequences of Srsf2 P95H mutation on hematopoiesis in transplantation settings. (A) The percentage of CD45.2+ donor cells in the PB of WT and KI mice. Data are shown as mean ± SD from 3 independent experiments. The numbers of mice analyzed are n = 27 at 8 weeks and n = 21 at 16 weeks after transplantation. (B) The percentage of CFSE-positive cells in the BM of WT and KI mice at 16 hours after transplantation (mean ± SD, n = 5). (C) Left panel: absolute number of nucleated BM cells in bilateral femurs and tibias from WT and KI mice. Right panel: Section of femurs stained with hematoxylin and eosin. Scale bars, 400 μm. (D-H) Total cell number of donor-derived CD45.2+ CD34−KSLs and CD34+KSLs (panel D); KSL cells (panel E); LT-HSCs, MPPs, HPC-1, and HPC-2 (panel F); myeloid progenitors (panel G); and CLPs (panel H) in the BM (mean ± SD, n = 7). (I) Cell cycle analysis in donor-derived LT-HSCs and KSL cells using Ki-67 and Hoechst 33342 staining (mean ± SD, n = 6). (J) Percentage of apoptotic cells in donor-derived LT-HSCs and KSL cells (mean ± SD: WT, n = 10; KI, n = 9). (K) Percentages of myeloid (Gr-1+ and/or Mac-1+), B-lymphoid (B220+), and T-lymphoid (CD4+ and/or CD8+) cells in the donor-derived CD45.2+ nucleated BM cells (mean ± SD, n = 7). (L) Percentages of pre-pro B (CD19+ B220+ IgM−), immature B (CD19+ B220mid IgM+), and mature B (CD19+ B220high IgM+) populations in the donor-derived CD45.2+ nucleated BM cells (mean ± SD, n = 7). In panels C through L, mice were analyzed at 15 to 20 weeks after transplantation. *P < .05; **P < .01; ***P < .001; ****P < .0001. IgM, immunoglobulin M. KI, Srsf2 mutant transplanted mice; WT, wild-type transplanted mice.
Consequences of Srsf2 P95H mutation on hematopoiesis in transplantation settings. (A) The percentage of CD45.2+ donor cells in the PB of WT and KI mice. Data are shown as mean ± SD from 3 independent experiments. The numbers of mice analyzed are n = 27 at 8 weeks and n = 21 at 16 weeks after transplantation. (B) The percentage of CFSE-positive cells in the BM of WT and KI mice at 16 hours after transplantation (mean ± SD, n = 5). (C) Left panel: absolute number of nucleated BM cells in bilateral femurs and tibias from WT and KI mice. Right panel: Section of femurs stained with hematoxylin and eosin. Scale bars, 400 μm. (D-H) Total cell number of donor-derived CD45.2+ CD34−KSLs and CD34+KSLs (panel D); KSL cells (panel E); LT-HSCs, MPPs, HPC-1, and HPC-2 (panel F); myeloid progenitors (panel G); and CLPs (panel H) in the BM (mean ± SD, n = 7). (I) Cell cycle analysis in donor-derived LT-HSCs and KSL cells using Ki-67 and Hoechst 33342 staining (mean ± SD, n = 6). (J) Percentage of apoptotic cells in donor-derived LT-HSCs and KSL cells (mean ± SD: WT, n = 10; KI, n = 9). (K) Percentages of myeloid (Gr-1+ and/or Mac-1+), B-lymphoid (B220+), and T-lymphoid (CD4+ and/or CD8+) cells in the donor-derived CD45.2+ nucleated BM cells (mean ± SD, n = 7). (L) Percentages of pre-pro B (CD19+ B220+ IgM−), immature B (CD19+ B220mid IgM+), and mature B (CD19+ B220high IgM+) populations in the donor-derived CD45.2+ nucleated BM cells (mean ± SD, n = 7). In panels C through L, mice were analyzed at 15 to 20 weeks after transplantation. *P < .05; **P < .01; ***P < .001; ****P < .0001. IgM, immunoglobulin M. KI, Srsf2 mutant transplanted mice; WT, wild-type transplanted mice.
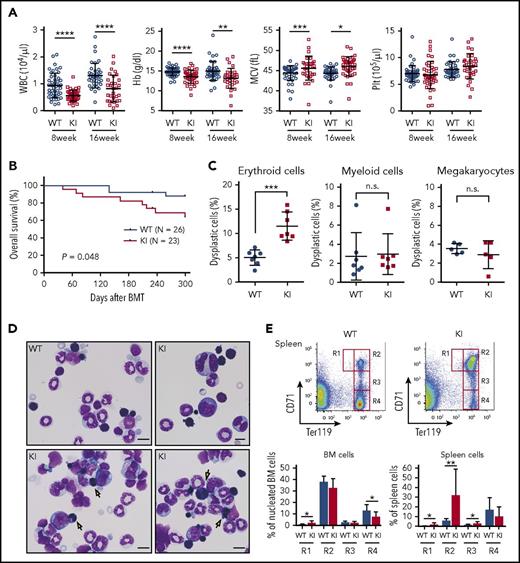
The mutant transplanted mice also showed additional evidence of abnormal dysplastic hematopoiesis. Compared with the control transplanted mice, the mutant transplanted mice had lower white blood cell counts and macrocytic anemia and showed a significantly shorter survival time with no evidence of leukemic transformation, which suggests BM failure as the major cause of deaths (Figure 4A-B; supplemental Figure 2A). The mutant transplanted animals also exhibited a slight but significant increase in the number of dysplastic erythroid cells in the BM, including those with multiple nuclei, compared with the wild-type transplanted mice (Figure 4C-D). Flow cytometry of the spleen and BM cells based on CD71 and Ter119 expression38 showed impaired erythroid differentiation (Figure 4E). The mutant transplanted mice demonstrated increased megakaryocytes in the spleen and increased paratrabecular megakaryocytes in the BM, which are characteristic of human MDS,39 indicating the presence of dysplastic megakaryopoiesis (supplemental Figure 2B-C).
Abnormal dysplastic hematopoiesis in Srsf2 mutant-transplanted mice. (A) Complete blood cell (CBC) counts of the PB from WT and KI mice are plotted as dots. The mean ± SD are presented as bars. Data were obtained from 4 independent experiments. The numbers of mice analyzed were n = 45 (WT) and n = 41 (KI) at 8 weeks and n = 38 (WT) and 31 (KI) at 16 weeks after transplantation. (B) Kaplan-Meier survival curve of WT (n = 26) and KI (n = 23) mice at 300 days after transplantation. (C) Percentages of dysplastic erythroid cells, myeloid cells, and megakaryocytes are plotted; bars indicate mean ± SD. In total, 7 pairs of WT and KI mice were analyzed for morphological evaluation of erythroid and myeloid cells, and 5 pairs were analyzed for morphological evaluation of megakaryocytes. For each mouse, >200 erythroid cells, >100 myeloid cells, and >15 megakaryocytes were counted. (D) Representative May–Grünwald–Giemsa staining of BM cells is shown; arrows point to binucleated erythroid cells. Magnification 1000×; scale bar, 10 μm. (E) Upper panels: representative flow cytometry analysis of erythroid lineage in the spleen, showing CD71highTer119med (R1), CD71highTer119high (R2), CD71medTer119high (R3), and CD71lowTer119high (R4) gates. Lower panels: percentage of erythroid precursors in the BM and spleen cells (mean ± SD, n = 7). *P < .05; **P < .01; ***P < .001; ****P < .0001. BMT, bone marrow transplantation. KI, Srsf2 mutant transplanted mice; WT, wild-type transplanted mice.
Abnormal dysplastic hematopoiesis in Srsf2 mutant-transplanted mice. (A) Complete blood cell (CBC) counts of the PB from WT and KI mice are plotted as dots. The mean ± SD are presented as bars. Data were obtained from 4 independent experiments. The numbers of mice analyzed were n = 45 (WT) and n = 41 (KI) at 8 weeks and n = 38 (WT) and 31 (KI) at 16 weeks after transplantation. (B) Kaplan-Meier survival curve of WT (n = 26) and KI (n = 23) mice at 300 days after transplantation. (C) Percentages of dysplastic erythroid cells, myeloid cells, and megakaryocytes are plotted; bars indicate mean ± SD. In total, 7 pairs of WT and KI mice were analyzed for morphological evaluation of erythroid and myeloid cells, and 5 pairs were analyzed for morphological evaluation of megakaryocytes. For each mouse, >200 erythroid cells, >100 myeloid cells, and >15 megakaryocytes were counted. (D) Representative May–Grünwald–Giemsa staining of BM cells is shown; arrows point to binucleated erythroid cells. Magnification 1000×; scale bar, 10 μm. (E) Upper panels: representative flow cytometry analysis of erythroid lineage in the spleen, showing CD71highTer119med (R1), CD71highTer119high (R2), CD71medTer119high (R3), and CD71lowTer119high (R4) gates. Lower panels: percentage of erythroid precursors in the BM and spleen cells (mean ± SD, n = 7). *P < .05; **P < .01; ***P < .001; ****P < .0001. BMT, bone marrow transplantation. KI, Srsf2 mutant transplanted mice; WT, wild-type transplanted mice.
Compromised reconstitution capacity of Srsf2-mutated stem cells
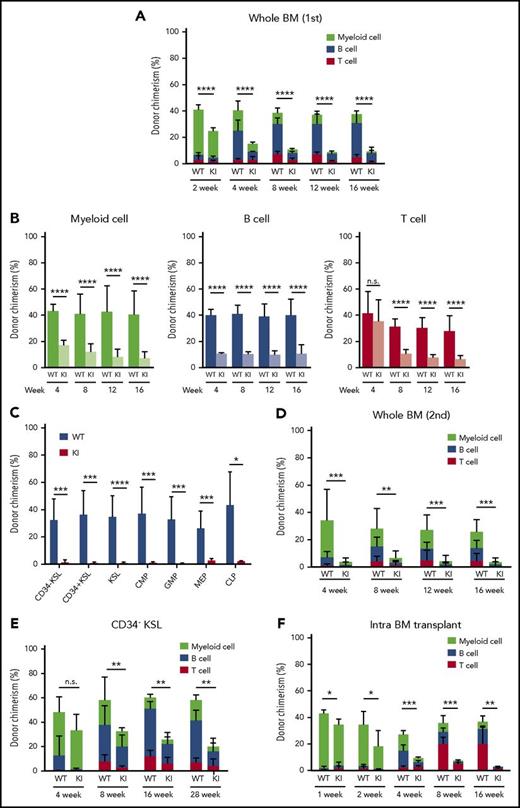
To further characterize Srsf2-mutated stem cells, we evaluated the reconstitution capacity of these HSCs using competitive repopulation assays. Compared with the wild-type transplanted mice, the mutant transplanted mice showed a significant and progressive reduction in the chimerism of donor-derived cells in PB, which was seen in all lineages examined (Figure 5A-B). At 4 months after transplantation, the chimerism of mutant-derived cells in the BM HSPC fractions declined to very low levels compared with that of wild-type–derived cells (Figure 5C). We next performed serial transplantation assays to evaluate the repopulating capacity of highly primitive Srsf2-mutated HSCs, in which the chimerism of mutant-derived cells in PB further declined (Figure 5D). The reduced repopulating capacity of mutant-derived stem cells was also demonstrated with use of a highly purified fraction of CD34−KSL HSCs (Figure 5E). To exclude a possibility of defective BM homing of mutant stem cells as the cause of reduced chimerism, we also performed an intra-BM competitive repopulation assay. We still observed the reduced chimerism of mutant-derived cells as early as 1 week after transplantation (Figure 5F), which supports the interpretation that the reduced chimerism of Srsf2-mutated HSCs was due to their impaired repopulating capacity.
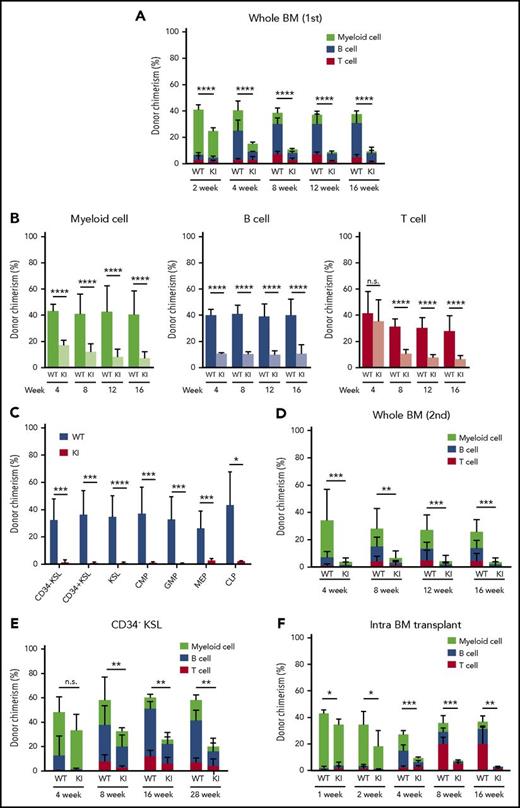
Compromised reconstitution capacity of Srsf2-mutated stem cells. (A) Percentage of CD45.2 donor cells in the PB of CD45.1 recipient mice after competitive transplantation using whole BM (mean ± SD, n = 10). (B) Chimerism of CD45.2 donor cells in myeloid (CD11b+ and/or Gr-1+), B-lymphoid (B220+), and T-lymphoid (CD4+ and/or CD8+) cells in PB (mean ± SD, n = 10). (C) Donor chimerism of stem and progenitor cell fractions in BM 18 weeks after competitive BMT (mean ± SD, n = 7). (D) Percentage of CD45.2 donor cells in PB of CD45.1 recipients after secondary transplantation (mean ± SD, n = 10). (E) Percentage of CD45.2 donor cells in PB after competitive transplantation using enriched LT-HSCs (CD34−KSL cells) (mean ± SD, n = 7). (F) Donor chimerism in PB after intra-BMT, indicated as mean ± SD (WT, n = 4; KI, n = 6). All of the transplantation experiments were performed in biological duplicate, and representative data are shown. *P < .05; **P < .01; ***P < .001; ****P < .0001. KI, Srsf2 mutant transplanted mice; WT, wild-type transplanted mice.
Compromised reconstitution capacity of Srsf2-mutated stem cells. (A) Percentage of CD45.2 donor cells in the PB of CD45.1 recipient mice after competitive transplantation using whole BM (mean ± SD, n = 10). (B) Chimerism of CD45.2 donor cells in myeloid (CD11b+ and/or Gr-1+), B-lymphoid (B220+), and T-lymphoid (CD4+ and/or CD8+) cells in PB (mean ± SD, n = 10). (C) Donor chimerism of stem and progenitor cell fractions in BM 18 weeks after competitive BMT (mean ± SD, n = 7). (D) Percentage of CD45.2 donor cells in PB of CD45.1 recipients after secondary transplantation (mean ± SD, n = 10). (E) Percentage of CD45.2 donor cells in PB after competitive transplantation using enriched LT-HSCs (CD34−KSL cells) (mean ± SD, n = 7). (F) Donor chimerism in PB after intra-BMT, indicated as mean ± SD (WT, n = 4; KI, n = 6). All of the transplantation experiments were performed in biological duplicate, and representative data are shown. *P < .05; **P < .01; ***P < .001; ****P < .0001. KI, Srsf2 mutant transplanted mice; WT, wild-type transplanted mice.
Effect of the Srsf2 P95H mutation on RNA splicing in vivo
We next evaluated the effect of mutant Srsf2 on RNA splicing through transcriptome sequencing of KSL cells and MPs (c-Kit+Sca-1-Linlow) purified from the BM of Vav1-Cre+/−Srsf2floxP95H/WT and Vav1-Cre+/−Srsf2WT/WT mice, as well as mutant and wild-type transplanted mice. By using the rMATS algorithm21 with a cutoff value of a false discovery rate (FDR) <0.1% and >5% for inclusion level differences, we identified 1484 and 1087 abnormal AS events in KSL and MP cells in transplantation settings, respectively, and 927 and 276 abnormal AS events in KSL and MP cells in the steady state, respectively. The types of abnormal AS events were consistent among all of the populations, in which the CEs were most frequently affected, followed by mutually exclusive exons, alternative 3′/5′ splice sites, and intron retentions. Srsf2 mutations were associated with a mild bias toward exclusion of CEs, alternative splice sites, and intron retentions (Figure 6A-B; supplemental Figure 3A; supplemental Table 2). The magnitude of mutant-associated splicing changes remained at less than 20% of authentic splicing reads for most of the differentially spliced events (>70%) (supplemental Figure 3B). The validity of the detection was confirmed by quantitative reverse transcription polymerase chain reaction for several splicing junctions (supplemental Figure 4).
Effect of Srsf2 P95H mutation on RNA splicing and expression in vivo. (A) Number of abnormal AS events identified in KSL cells of KI mice compared with that of WT mice in transplantation settings (n = 6 pairs). The log10 ratio of the number of abnormal AS events differentially included to those differentially excluded is also shown. (B) Inclusion levels of the CEs and mutually exclusive exons in KSL cells of KI mice compared with that of WT mice in transplantation settings are plotted. The colored dots represent significantly more included (red) and excluded (blue) events. The gray dots represent nonsignificant AS events with ≤5% inclusion level differences. (C) The composition of SSNG motifs in the more included or excluded CEs in the KSL cells of KI mice compared with that of WT mice in transplantation settings, and SSNG motif compositions in all of the exons from the mouse reference genome (mm10). (D) Relative frequency (log10 ratio) of the 7-base window-averaged number of CCNG and GGNG motifs in the CEs promoted vs repressed in KSL cells of KI mice, extending 100 nt into their upstream and downstream from the 5′ and 3′ splice sites. (E) Venn diagram comparing the differentially spliced genes between KI and WT cells from 4 different populations, ie, KSL and MP cells from the steady-state mice and those from the recipient mice in transplantation settings. Genes registered in the Cancer Gene Census are shown in red. (F) MA plot and volcano plot showing the transcriptional changes between KI and WT KSL cells in transplantation settings. Significantly upregulated and downregulated genes in KI cells compared with control cells are indicated by red and blue, respectively. (G) Venn diagram comparing the differentially upregulated (left) and downregulated (right) genes in Srsf2 mutant KSL and MP cells from the steady-state mice vs those from the recipient mice in transplantation settings. Genes registered in the Cancer Gene Census are shown in red. (H) Gene set enrichment analysis demonstrating a significant positive enrichment of gene sets upregulated in human MDS compared with healthy controls in Srsf2 mutant KSL cells relative to wild-type KSL cells in transplantation settings (left panel), and negative enrichment of gene sets associated with the stemness (middle panel) and quiescence (right panel) in Srsf2 mutant KSL cells relative to wild-type KSL cells under the steady-state conditions. Nominal P values, FDRs, and normalized enrichment scores (NSEs) are indicated. KI, Srsf2 mutant transplanted mice; WT, wild-type transplanted mice.
Effect of Srsf2 P95H mutation on RNA splicing and expression in vivo. (A) Number of abnormal AS events identified in KSL cells of KI mice compared with that of WT mice in transplantation settings (n = 6 pairs). The log10 ratio of the number of abnormal AS events differentially included to those differentially excluded is also shown. (B) Inclusion levels of the CEs and mutually exclusive exons in KSL cells of KI mice compared with that of WT mice in transplantation settings are plotted. The colored dots represent significantly more included (red) and excluded (blue) events. The gray dots represent nonsignificant AS events with ≤5% inclusion level differences. (C) The composition of SSNG motifs in the more included or excluded CEs in the KSL cells of KI mice compared with that of WT mice in transplantation settings, and SSNG motif compositions in all of the exons from the mouse reference genome (mm10). (D) Relative frequency (log10 ratio) of the 7-base window-averaged number of CCNG and GGNG motifs in the CEs promoted vs repressed in KSL cells of KI mice, extending 100 nt into their upstream and downstream from the 5′ and 3′ splice sites. (E) Venn diagram comparing the differentially spliced genes between KI and WT cells from 4 different populations, ie, KSL and MP cells from the steady-state mice and those from the recipient mice in transplantation settings. Genes registered in the Cancer Gene Census are shown in red. (F) MA plot and volcano plot showing the transcriptional changes between KI and WT KSL cells in transplantation settings. Significantly upregulated and downregulated genes in KI cells compared with control cells are indicated by red and blue, respectively. (G) Venn diagram comparing the differentially upregulated (left) and downregulated (right) genes in Srsf2 mutant KSL and MP cells from the steady-state mice vs those from the recipient mice in transplantation settings. Genes registered in the Cancer Gene Census are shown in red. (H) Gene set enrichment analysis demonstrating a significant positive enrichment of gene sets upregulated in human MDS compared with healthy controls in Srsf2 mutant KSL cells relative to wild-type KSL cells in transplantation settings (left panel), and negative enrichment of gene sets associated with the stemness (middle panel) and quiescence (right panel) in Srsf2 mutant KSL cells relative to wild-type KSL cells under the steady-state conditions. Nominal P values, FDRs, and normalized enrichment scores (NSEs) are indicated. KI, Srsf2 mutant transplanted mice; WT, wild-type transplanted mice.
SRSF2 is known to promote inclusion or exclusion of exons through binding to specific exonic splicing enhancers,6,40,41 in which 5′-SSNG-3′ (S=C/G, N=A/U/C/G) consensus motifs provide high-affinity binding sites.8 Based on this knowledge, we investigated 4-mers that were significantly enriched in the CEs differentially spliced in Srsf2-mutated cells. Consistent with previous reports,12,13 CCNG and GGNG were significantly enriched in those exons that were more included (splicing promoted) and excluded (splicing repressed) in the Srsf2-mutated cells, respectively, whereas no significantly different features in the intron-exon boundary sequences were identified between Srsf2P95H-promoted and Srsf2P95H-repressed exons (Figure 6C; supplemental Figure 3C-D). CCNG motifs tended to be slightly enriched in ∼20 bp from the 3′ splice site of promoted exons compared with more distal upstream intronic regions, whereas GGNG motifs were enriched over the repressed CEs compared with their surrounding intronic regions (Figure 6D; supplemental Figure 3E).
Functional targets of the Srsf2 P95H mutation
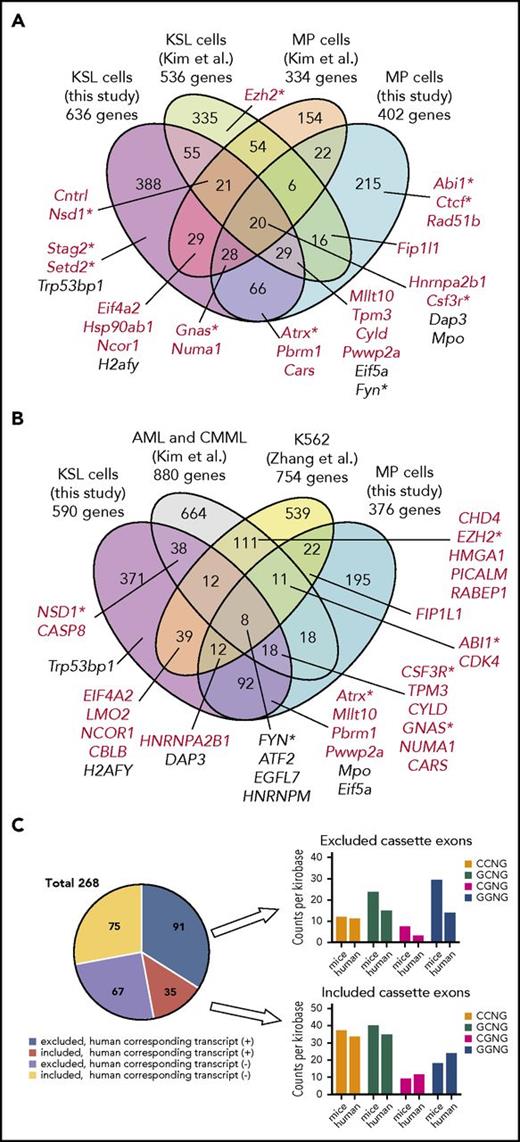
To identify the potential functional targets of mutant Srsf2, we interrogated differentially spliced genes between mutant and wild-type Srsf2 for KSL and MP fractions in both the steady-state and transplantation settings. We identified a total of 1189 differentially spliced genes in at least one of the 4 fractions (Figure 6E). Among these, 98 genes were differentially spliced in more than 3 populations, of which 13 were registered in the Cancer Gene Census (http://cancer.sanger.ac.uk/census), such as Hnrnpa2b1, Gnas, Atrx, Csf3r, and Mllt10 (Table 1). Higher numbers of abnormal AS events were observed in KSL and MP cells from Srsf2 mutant transplanted mice, compared with those from steady-state mice. These AS events affected several genes implicated in leukemogenesis (Stag2, Bap1, and Ctcf), DNA damage response (Chek2, Fancc, Rad51b, and Cdk4), and apoptosis (Casp8) (Figure 6E). We also explored those genes showing significant changes in gene expression levels between Srsf2 mutant and wild-type and identified 387 upregulated and 548 downregulated genes, with FDR <0.05 and >1.2-fold change, in at least one of the 4 populations, including Hnrnpa2b1 and Gata1 (upregulated) and Mpl, Irf4, Csf3r, and Trp53bp1 (downregulated) (Figure 6F-G; supplemental Figure 3F; supplemental Table 3). Gene set enrichment analysis42,43 revealed significant enrichment of pathways deregulated in human MDS44 in Srsf2 mutant KSL cells of transplanted recipients but not in those of nontransplanted mice. Srsf2 mutant HSPCs also showed significant enrichment of several pathways, including loss of stemness, enhanced cell cycle and DNA repair, impaired differentiation, and upregulation of the genes involved in RNA metabolism both in the steady-state and transplantation settings (Figure 6H; supplemental Table 4). We detected no statistically significant overlaps between the differentially expressed genes and differentially spliced genes in any of the 4 cell fractions. Although only a small number of misspliced genes were predicted to be degraded by the nonsense-mediated decay pathway among the downregulated genes (supplemental Figure 5), several genes showed significant changes in both RNA splicing and expression, including Hnrnpa2b1, Csf3r, and Trp53bp1.
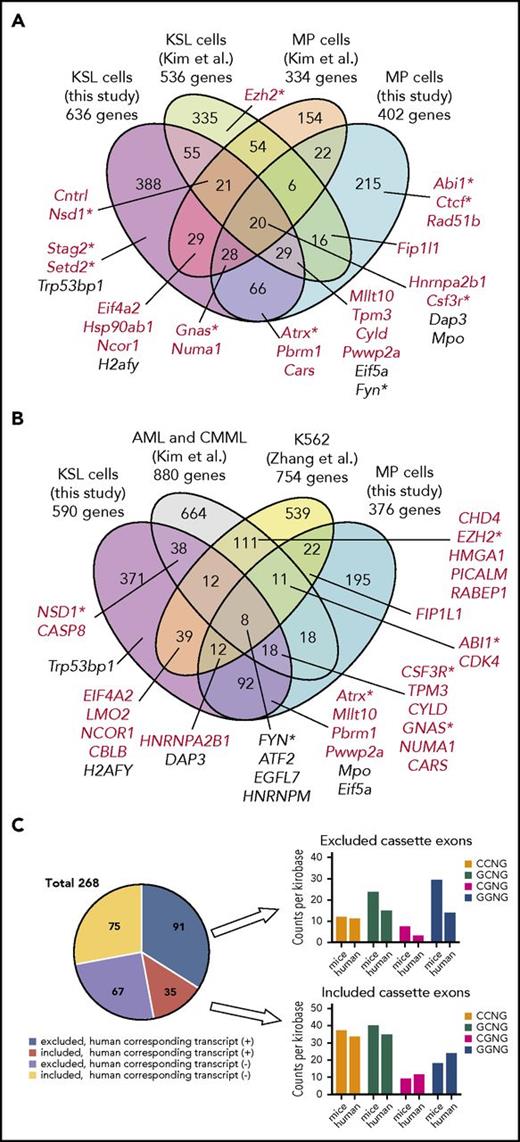
We next compared our results with those obtained from previous studies in a different mouse model and a human cell line.12,13 To minimize potential biases from the analysis platform, we reanalyzed original RNA-seq data from a Mx1-Cre Srsf2P95H mouse model12 and SRSF2 mutant K562 cells13 using the same algorithm (rMATS) and cutoff points that we adopted for the analysis of our mouse model (supplemental Table 5). There were significant overlaps between the targets detected in our study compared with those of previous studies, which support the validity of the our current result. Among the 895 genes undergoing abnormal AS in KSL and/or MP cells in transplantation settings in our mouse model, 147 and 126 were also identified in the analysis of KSL cells (P < 10−6) and MP cells (P < 10−6) from different Srsf2-mutated mouse strains,12 respectively (Figure 7A). Also, among the misspliced genes in KSL and/or MP cells in transplantation settings, 105 and 104 genes were overlapped with the targets identified in SRSF2-mutated primary human leukemia samples (P < 10−6)12 and a cell line transduced with mutant SRSF2 (P < 10−6),13 respectively (Figure 7B). In addition to the previously reported targets, we newly identified a number of novel target genes of abnormal AS events, such as Stag2 and Setd2. Genes that were differentially spliced in both mutant mouse strains and human samples included several genes implicated in the pathogenesis of myeloid malignancies; these include Csf3r, Fyn, Gnas, and Nsd1.45-49 Among the CEs altered in both KSL and MP cells but not in human samples, approximately half had corresponding transcripts in the human orthologs; for those CEs, the frequency of CCNG motifs within the promoted CEs and GGNG motifs within the repressed CEs was decreased in human counterparts, which may in part account for the different splicing patterns between species (Figure 7C).
Comparisons of misspliced genes in Srsf2-mutated cells between different mouse models and human cells. (A) Comparisons of misspliced genes in KSL and MP cells from Srsf2 mutant transplanted mice in the current study and those from Mx1-Cre–mediated Srsf2 mutant mice (Kim et al12 ). Genes registered in the Cancer Gene Census are shown in red. Genes implicated in the pathogenesis of myeloid malignancies are marked with asterisks. (B) Comparisons of differentially spliced genes in KSL and MP cells from Srsf2 mutant transplanted mice in the present study, SRSF2-mutated primary acute myeloid leukemia (AML) and CMML samples (Kim et al12 ), and K562 cell line transduced with mutant SRSF2 (Zhang et al13 ). Genes registered in the Cancer Gene Census are shown in red. Genes implicated in the pathogenesis of myeloid malignancies are marked with asterisks. (C) Pie chart showing the number of promoted/repressed CEs in both KSL and MP cells from Srsf2 mutant transplanted mice but not in SRSF2 mutant human samples, with or without corresponding transcripts of the human orthologs. For those CEs who had human corresponding transcripts, the frequency of SSNG motifs (counts per kilobase) was compared between mice and humans.
Comparisons of misspliced genes in Srsf2-mutated cells between different mouse models and human cells. (A) Comparisons of misspliced genes in KSL and MP cells from Srsf2 mutant transplanted mice in the current study and those from Mx1-Cre–mediated Srsf2 mutant mice (Kim et al12 ). Genes registered in the Cancer Gene Census are shown in red. Genes implicated in the pathogenesis of myeloid malignancies are marked with asterisks. (B) Comparisons of differentially spliced genes in KSL and MP cells from Srsf2 mutant transplanted mice in the present study, SRSF2-mutated primary acute myeloid leukemia (AML) and CMML samples (Kim et al12 ), and K562 cell line transduced with mutant SRSF2 (Zhang et al13 ). Genes registered in the Cancer Gene Census are shown in red. Genes implicated in the pathogenesis of myeloid malignancies are marked with asterisks. (C) Pie chart showing the number of promoted/repressed CEs in both KSL and MP cells from Srsf2 mutant transplanted mice but not in SRSF2 mutant human samples, with or without corresponding transcripts of the human orthologs. For those CEs who had human corresponding transcripts, the frequency of SSNG motifs (counts per kilobase) was compared between mice and humans.
Previously, Kim et al12 reported that SRSF2 mutant cells exhibited preferential inclusion of a poison CE that introduces a premature termination codon predicted to trigger nonsense-mediated decay of EZH2. However, in the present study, we did not detect those CEs whose splicing pattern was significantly different between wild-type and mutant cells, not only in HSPCs but in more differentiated fractions, including what corresponds to the exon reported to be increased in human EZH2 and in those reported to be changed in mice (supplemental Figure 6A-B). No significant difference was detected in the Ezh2 protein levels either (supplemental Figure 6C). We found two CCNG motifs within the EZH2 poison CEs in human samples, in contrast to one CCNG motif in the mouse counterpart (supplemental Figure 6D), which may account for the discrepancy between the species.
Discussion
Effects of SF mutations on hematopoiesis were first examined by Yoshida et al,5 using competitive transplantation of purified mouse stem cells transduced with different U2AF1 mutants (S34F, Q157P, and Q157R). More recently, other groups have investigated Mx1-Cre–mediated conditional knockin mouse models of the Srsf2(P95H)12 and the Sf3b1(K700E)15,16 mutants, as well as a doxycycline-inducible transgenic mouse model for the U2Af1(S34F) mutant.14 In all of these studies, SF-mutated HSCs exhibited a compromised repopulation capacity compared with wild-type HSCs, which were also recapitulated in our Vav1-Cre–inducible Srsf2 mutant mice. Thus, one of the common consequences of Srsf2 and other SF mutations seem to be compromised HSC function in terms of repopulating capacity. Given that SF mutations are implicated in early genetic events in myeloid leukemogenesis3,4,11 and in clonal hematopoiesis in aged-normal populations (age-related clonal hematopoiesis [ARCH] or clonal hematopoiesis with indeterminate potential [CHIP]),50-54 this trend raises a perplexing question: How can these functionally compromised HSCs harboring SF mutations be clonally selected in the early stages in leukemogenesis and ARCH/CHIP? Differential effects of these mutations on aged and nonaged stem cells and/or an abnormal BM environment that specifically favors SF-mutated HSCs may be among possible explanations.
In accordance with the previous study reported by Kim et al,12 Srsf2 mutant transplanted mice developed an MDS-like phenotype, showing leukopenia, anemia, and dysplastic morphologic features. A similar phenotype was also found in the steady-state condition, although the phenotype was much milder: No dysplastic phenotypes were observed, and only a mild decrease in hemoglobin was detected. Although the short life span of mouse models could limit the accumulation of genetic and/or epigenetic damages required for MDS development, it has been demonstrated that transplantation exposes cells to replicative stress by inducing rapid cycling of dormant HSCs, which leads to the accumulation of DNA damage in HSCs in a way that resembles age-associated effects.35,36 Of interest is that higher numbers of abnormal AS events were observed in transplantation settings, where the targets of abnormal AS included those genes implicated in leukemogenesis, DNA damage response, and apoptosis. These findings suggest that mutant Srsf2-related abnormal AS is augmented by replicative stress, contributing to the enhanced MDS-like phenotype in Srsf2 mutant mice in transplantation settings. This is also of interest in view of elderly onset of MDS and SF mutations associated with ARCH/CHIP.50,51
In sharp contrast to the previous study by Kim et al,12 in which they reported increased numbers of HSPCs associated with mutant Srsf2, we found substantially reduced numbers of immature HSPCs both in the steady-state mouse BM and in the setting of transplantation. The reduced numbers of HSCs in our study, which were confirmed by limiting dilution experiments, seem to be in accordance with a reduced competitive repopulation capacity, increased cell cycling, and enhanced apoptosis, which might result in stem cell depletion. The precise reason for the differences between the present study and the study by Kim et al,12 however, is still elusive. The effect of mutant Srsf2 on the number of stem cells might be context dependent, in which the differences in the mutant construct (FLEx switch vs loxP-cDNA-STOP-loxP-mut cDNA), the Cre strain (Vav1-Cre vs Mx1-Cre) used for expression of the mutant allele, and/or the timing of mutant Srsf2 expression in transplantation settings (before transplantation in our study; after transplantation in the study by Kim et al12 ) could be plausible explanations.
The functional targets of abnormal splicing responsible for the phenotype of Srsf2-mutated hematopoiesis are still elusive. Alternative CE usage should cause deletions or insertions of corresponding amino acids, likely resulting in loss of function of the protein in most cases, although the precise consequence is not necessarily easy to predict. As shown beforehand, the consensus-binding motifs were substantially different between human and mouse, which might partly explain the different splicing patterns between the 2 species. Nevertheless, significant overlaps of the misspliced genes in Srsf2-mutated cells still existed between different mouse models and human cells, in which a number of genes implicated in human hematopoietic malignancies were affected; these included Csf3r, Fyn, Gnas, Nsd1, and Hnrnpa2b1. In addition, several genes underwent significant changes in both RNA splicing and expression between Srsf2 mutant and wild-type cells, including Hnrnpa2b1, Csf3r, and Trp53bp1, which could be the candidates of functional targets of Srsf2 mutation. Meanwhile, it is also unclear whether all of the hematological phenotypes associated with Srsf2 mutants could be explained by abnormal splicing in a particular gene or set of genes. It might be also possible that a large number of abnormally spliced transcripts and consequent abnormal proteins, irrespective of the particular genes involved, cause compromised stem cell functions, contributing to MDS phenotypes. Further studies regarding these issues should be warranted.
In conclusion, the present study described the in vivo effects of the mutant Srsf2 allele on the phenotypes of steady-state hematopoiesis, which were augmented in transplantation settings. Srsf2 P95H mutation can cause abnormal RNA splicing typically of CEs and induce deregulation of HSCs, but it may not be sufficient to recapitulate a full picture of MDS at least in the steady-state condition. Our mouse model provides a valuable tool to understand the molecular pathogenesis of Srsf2-mutated myeloid neoplasms.
The sequencing data reported in this article have been deposited in the DNA Data Bank of Japan Sequence Read Archive (accession number DRA006224).
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
The authors thank Yuji Yamazaki for expert technical assistance in the fluorescence-activated cell-sorting operation; Hiroko Tsukui, Naomi Sato, Satoko Yabuta, Ai Takatsu, and Maki Nakamura for technical assistance; and Maiko Nakamura for precious secretarial assistance.
This work was supported by a Grant-in-Aid for Scientific Research (MEXT/JSPS KAKENHI JP26221308 and JP26253060 to S.O., JP16K19574 and JP14J03784 to A.K., and JP26115009 to M.S.); a Grant-in-Aid from the Japan Agency for Medical Research and Development (Project for Development of Innovative Research on Cancer Therapeutics [16cm0106501h0001 to S.O.], Practical Research for Innovative Cancer Control Project [15ck0106073h0002 and 16ck0106073h0003 to S.O.], and Strategic International Collaborative Research Program [16jm0210034h0104 to H.N.]); and grants from the Takeda Science Foundation (S.O.) and the Uehara Memorial Foundation (A.K.).
Authorship
Contribution: A.K. designed the study, analyzed the data, and wrote the manuscript; A.K. and S.Y. performed the animal experiments; A.K., M.S., and K.Y. performed the RNA-seq experiments; Y.N. and Y.S. performed bioinformatics analyses of the sequencing data; A.K. and Y.O. performed histopathological analyses; K.K. and M.M.N. provided the scientific discussion; M.M. and T.Y. assisted with the experiments; M.N. and H.K. generated Srsf2 conditional knockin mice; H.N. and S.O. jointly supervised the project; and S.O. conceived and led the project, and wrote the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Hiromitsu Nakauchi, Institute for Stem Cell Biology and Regenerative Medicine, Stanford University School of Medicine, 265 Campus Dr, Stanford, CA 94305; e-mail: nakauchi@stanford.edu; and Seishi Ogawa, Department of Pathology and Tumor Biology, Graduate School of Medicine, Kyoto University, Yoshida-Konoe-cho, Sakyo-ku, Kyoto-shi, Kyoto 606-8501, Japan; e-mail: sogawa-tky@umin.ac.jp.
REFERENCES
Author notes
H.N. and S.O. contributed equally to this study.