Key Points
Dose- and time-dependent effects of siFga allow tailored fibrinogen knockdown.
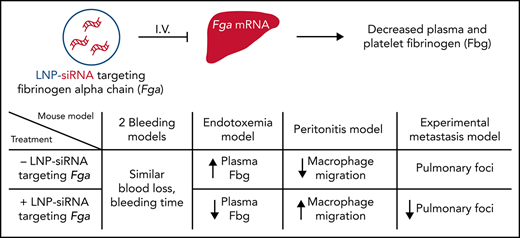
Administration of LNP-encapsulated siFga is a promising strategy to modulate multiple fibrin(ogen)-dependent pathologies.
Abstract
Fibrinogen plays a pathologic role in multiple diseases. It contributes to thrombosis and modifies inflammatory and immune responses, supported by studies in mice expressing fibrinogen variants with altered function or with a germline fibrinogen deficiency. However, therapeutic strategies to safely and effectively tailor plasma fibrinogen concentration are lacking. Here, we developed a strategy to tune fibrinogen expression by administering lipid nanoparticle (LNP)-encapsulated small interfering RNA (siRNA) targeting the fibrinogen α chain (siFga). Three distinct LNP-siFga reagents reduced both hepatic Fga messenger RNA and fibrinogen levels in platelets and plasma, with plasma levels decreased to 42%, 16%, and 4% of normal within 1 week of administration. Using the most potent siFga, circulating fibrinogen was controllably decreased to 32%, 14%, and 5% of baseline with 0.5, 1.0, and 2.0 mg/kg doses, respectively. Whole blood from mice treated with siFga formed clots with significantly decreased clot strength ex vivo, but siFga treatment did not compromise hemostasis following saphenous vein puncture or tail transection. In an endotoxemia model, siFga suppressed the acute phase response and decreased plasma fibrinogen, D-dimer, and proinflammatory cytokine levels. In a sterile peritonitis model, siFga restored normal macrophage migration in plasminogen-deficient mice. Finally, treatment of mice with siFga decreased the metastatic potential of tumor cells in a manner comparable to that observed in fibrinogen-deficient mice. The results indicate that siFga causes robust and controllable depletion of fibrinogen and provides the proof-of-concept that this strategy can modulate the pleiotropic effects of fibrinogen in relevant disease models.
Introduction
Fibrinogen is synthesized by the liver and circulates in plasma at a concentration of 2 to 4 g/L, with a half-life of 3 to 5 days in plasma.1 Fibrinogen contributes to multiple pathologies by modifying inflammatory and malignant processes.1-4 Depending on the context, the immunomodulatory effects of fibrin(ogen) may be beneficial or detrimental. Extravascular fibrin(ogen) deposits at sites of tissue damage recruit leukocytes to limit pathogen dissemination and promote pathogen clearance and contribute to tissue repair.4-6 In contrast, fibrin(ogen) contributes to the metastasis of tumor cells by inhibiting the activity of natural killer cells.7,8 The hepatic expression of fibrinogen is significantly upregulated during acute phase response to inflammatory challenges, such as in COVID-19,9,10 cancer,11,12 sepsis,13,14 and obesity.15,16 Although fibrinogen is essential for hemostasis, elevated fibrinogen (hyperfibrinogenemia) is an independent risk factor for thrombosis, through increased blood viscosity and resistance to fibrinolysis.2,10,17
Controllably decreasing circulating fibrinogen without compromising hemostasis has both experimental and therapeutic applications. However, strategies to safely decrease the concentration of fibrinogen in plasma for long durations do not exist. Fibrinogen-depleting proteases isolated from snake venom have been used for reperfusion therapy on rare occasions, but their efficacy has not been clearly demonstrated.18,19 This treatment also results in increased levels of circulating fibrinogen degradation products, which also contribute to inflammation.4,18,20 Single-stranded antisense oligonucleotides (ASOs) decrease circulating fibrinogen in mice.21 However, as a therapeutic platform, ASOs face broad challenges to clinical use, including liver and kidney toxicity, and thrombocytopenia.22 These effects likely occur as a result of frequent administration of large doses of ASO to achieve sufficient messenger RNA (mRNA) knockdown. Long-term knockdown of mRNA and proteins using small interfering RNA (siRNA) typically requires lower doses of nucleic acid than ASOs, particularly when delivered using lipid nanoparticles (LNPs) to knock down mRNA in hepatocytes.23 Here, we tested the hypothesis that LNP delivery of siRNA targeting the fibrinogen α chain (Fga) mRNA could achieve controlled and sustained fibrinogen knock down in blood and modulate fibrin(ogen)-dependent disease processes.
Materials and methods
Preparing LNP-siRNA targeting mouse Fga
Three 2′O-methylated siRNA sequences targeting different regions of murine Fga mRNA (siFga) were designed in silico in consultation with Integrated DNA Therapeutics (IDT), using Smith-Waterman analysis to identify sequences that do not cross-react with other genes in the host transcriptome.24 An siRNA sequence targeting luciferase (siLuc) was used as negative control. Each siRNA sequence, synthesized by IDT, was packaged into LNPs with the same lipid composition as the first Food and Drug Administration–approved siRNA therapeutic, Onpattro, consisting of an ionizable cationic lipid (DLin-MC3-DMA), a phosphatidylcholine, cholesterol, and a polyethylene glycol lipid.25 siRNA-LNPs were produced and analyzed as previously described,26 then diluted to the desired concentration and administered to mice at the indicated dose. When siRNA-LNPs were used within a week of formulation, they were stored at 4°C, and dilutions were made with phosphate-buffered saline (PBS). If longer storage was needed, dilutions were made with a buffer containing 10 mM l-histidine and 10% sucrose (pH 7.4) and stored at −80°C until ready to use.
Preparing LNP-siRNA targeting human FGA
Five siRNA sequences targeting different regions of human FGA mRNA (hsiFga) were designed in silico in consultation with IDT as described above. Empty LNPs were formulated by combining lipids (a cationic lipid, a phosphatidylcholine, cholesterol, and a polyethylene glycol lipid) and 25 mM sodium acetate (pH 4.0) via microfluidic mixing, then dialyzing against 25 mM sodium acetate (pH 4.0). The cholesterol content of the empty LNPs was measured by using a Cholesterol E Assay Kit (Wako Chemicals, Mountain View, CA) to calculate the total lipid concentration. Empty LNPs and hsiFga were mixed at the desired concentrations and hsiFga:lipid ratios. The resulting LNP-siRNA was diluted to 3 μg/mL in Dulbecco′s Modified Eagle Medium (plus 10% heat inactivated fetal bovine serum) before transfecting the cells as described below.
Mice
Procedures performed at each institution were approved by the local Animal Care Committee, including The University of British Columbia (UBC), Michigan State University (MSU), University of North Carolina (UNC) at Chapel Hill, University of Cincinnati (Cincinnati Children's Hospital Medical Center), and National Institutes of Health (National Institute of Dental and Craniofacial Research). Wild-type (WT) C57BL/6J mice (The Jackson Laboratory) aged 8 to 14 weeks were used unless otherwise indicated.
Determination of plasma and platelet fibrinogen levels after siRNA treatment
Male and female WT mice were treated with siFga or siLuc (1 mg siRNA per kg body weight) via tail vein injection at UBC. Blood samples were collected 1 and 3 weeks later via intracardiac puncture into a syringe containing sodium citrate (0.32% final), and platelets and plasma fractions were isolated. Whole blood was centrifuged at 1500g for 10 minutes twice to obtain platelet-poor plasma. To isolate platelets, 300 μL Tyrode's buffer (pH 6.5) was added to whole blood and then centrifuged at 600g for 3 minutes to obtain platelet-rich plasma (PRP). Prostaglandin E1 (10 μg/mL; Sigma) was added to PRP and all subsequent spin steps to minimize platelet activation. PRP was centrifuged again at 400g for 2 minutes to remove remaining red blood cells. PRP was subsequently centrifuged at 800g for 10 minutes to isolate the pelleted platelets. Plasma fibrinogen was measured using a Mouse Total Fibrinogen ELISA kit (Innovative Research) or using a matched-pair antibody set for murine fibrinogen antigen (MFG-EIA; Enzyme Research Laboratories). To quantify platelet fibrinogen levels, western blotting was performed as previously described.26 In brief, samples were separated by sodium dodecyl–sulfate polyacrylamide gel electrophoresis and transferred to a nitrocellulose membrane (GE Healthcare). The membrane was incubated with rabbit anti-human fibrinogen antibody (1:10 000; A0080; Agilent Dako) or platelet factor 4 (PF4; 1:1000; SAPF4-AP; Affinity Biologicals). After incubating with horseradish peroxidase–conjugated goat anti-rabbit secondary antibody (1:20 000; ab7090; Abcam), specific bands were detected using enhanced chemiluminescence substrate (Bio-Rad) using a Sapphire Biomolecular Imager (Azure Biosystems). Quantification of fibrinogen bands was performed using ImageJ software and expressed as relative intensity to the PF4 loading control.
Determining hepatic fibrinogen knockdown after siRNA treatment
Liver tissue was excised at endpoint and further processed as previously described.26 Detection and quantification of fibrinogen gene expression by SYBR Green real-time polymerase chain reaction was performed using primers (IDT) against murine Fga (F: TGTGGAGAGACATCAGAGTCAATG, R: CGTCAATCAACCCTTTCATCC) and Ppia (F: GCGTCTCCTTCGAGCTGTT, R: TGTAAAGTCACCACCCTGGC) mRNA. Fibrinogen gene expression was quantified using the ΔΔCt method, relative to the expression of the housekeeping gene (Ppia).
Determining human FGA mRNA knockdown in human hepatoma (HUH7) cells transfected with hsiFga
HUH7 cells were seeded at 1 × 105 cells per well 1 day prior to transfection. Cells were then transfected with the 5 different LNP-hsiFga at a dose of 3 μg/mL of siRNA, or empty LNPs as a negative control. The following day, cells were lysed, and the RNA was extracted using the PureLink RNA Mini Kit (Thermo Fisher) following the manufacturer’s protocol. Fibrinogen gene expression was then detected and quantified using SYBR Green real-time polymerase chain reaction. Primers were designed against human Fga (F: GCTCTGATGAAGACTGGAA, R: ACGAATGAGAATCCTTATTGTTC) and actin (Act; F: CACCAACTGGGACGACAT, R: ACAGCCTGGATAGCAACG) mRNA. Fibrinogen gene expression was quantified using the ΔΔCt method, relative to the expression of the housekeeping gene (Act).
Determining suitability of frozen siRNA-LNP formulations
WT mice were injected retro-orbitally with either fresh siFga or frozen siFga at 1 mg/kg at MSU. Fresh siLuc (1 mg/kg) was used as control. Blood was collected under isoflurane anesthesia by exsanguination from the caudal vena cava into a syringe containing sodium citrate (0.38% final). Samples were centrifuged at 4000g for 10 minutes to obtain plasma. Plasma fibrinogen levels were determined as described above.
Dosing studies with siFga
Male WT mice were injected retro-orbitally with 0.1, 0.5, 1.0, or 2.0 mg/kg siFga at MSU. Blood was collected into sodium citrate (0.38% final) via retro-orbital bleed 3 days prior to siFga injections to obtain baseline fibrinogen plasma levels, and then once weekly after injection for a total of 4 weeks. Samples were processed, and plasma fibrinogen levels were determined as described above.
Thromboelastography (TEG) analysis
Coagulation and fibrinolytic properties of mouse whole blood were evaluated by TEG (Hemostasis Analyzer System 5000; Haemonetics) at 37°C at UBC. Citrated whole blood collected by intracardiac puncture was mixed with a calcium-saline buffer (50 mM CaCl2 and 90 mM NaCl) and recombinant tissue factor (Innovin; 10 pM; MedCorp).
Saphenous vein bleeding model
Male and female WT mice were injected with siFga (1 mg/kg) or vehicle control (PBS) via tail vein 1 week prior to the bleeding challenge at UBC. Mice were anesthetized by isoflurane inhalation. Saphenous vein bleeding was initiated and monitored in a simplified version of an injury model previously described.27 Briefly, the saphenous vein was isolated, and a puncture was made in the medial wall of the vein using a 23-gauge needle. Blood loss over time was measured by gently absorbing blood at the puncture site with preweighed filter paper until bleeding stopped. Bleeding was monitored for 40 minutes.
Tail transection bleeding model
Male and female WT mice were injected with siFga or siLuc (1 mg/kg) 1 week prior to the bleeding challenge at UBC. Mice were anesthetized by isoflurane inhalation. Tails were transected 4 mm from the tip and immediately immersed in a 0.9% NaCl solution (saline) to monitor bleeding for 20 minutes. To quantify blood loss, the blood-saline samples were treated with a red blood cell lysis solution (1.5 M NH4Cl, 0.1 M NaHCO3, 0.01 M EDTA), incubated at room temperature for 10 minutes while gently shaking; then, absorbance was measured at 509 nm (Tecan microplate reader). The absorbance was converted to amount of blood loss, using a standard curve with known amounts of mouse blood collected by intracardiac puncture, and normalized to body weight.
siFga in an endotoxemia model
Male WT mice were injected with siFga or siLuc (1 mg/kg) via tail vein 1 week prior to intraperitoneal injection with 10 mg/kg of lipopolysaccharide (LPS; Sigma) at UNC Chapel Hill. Mice were euthanized 24 hours after LPS injection, and blood and livers were collected for analysis. Hepatic mRNA expression levels of Fga, fibrinogen β chain (Fgb), and fibrinogen γ chain (Fgg) were determined using TaqMan gene expression assays (Applied Biosystems) on an ABI StepOne Plus sequence detection system (Applied Biosystems). The expression of each gene was normalized relative to β2-microglobulin (B2m) expression levels, and relative expression level was determined using the Pfaffl method.28 Plasma levels of fibrinogen, and D-dimer were quantified by enzyme-linked immunosorbent assay (ELISA; Immunology Consultants Laboratory Inc, Siemens Healthcare Diagnostics, and Diagnostica Stago, respectively). Cytokines in platelet-poor plasma, including tumor necrosis factor-α (TNF-α), interleukin-1β (IL-1β), IL-17, monocyte chemoattractant protein-1 (MCP-1), and macrophage inflammatory protein-1α (MIP-1α), were measured using a multiplex cytokine analysis by the Advanced Analytics Core at UNC Chapel Hill.
siFga in thioglycollate-induced peritonitis
Male and female plasminogen-deficient (Plg−/−) mice were injected with siFga or siLuc (1 mg/kg) 1 week prior to inducing peritonitis at the National Institute of Dental and Craniofacial Research. Plasminogen-sufficient (Plg+/+) mice were not treated with siRNA prior to peritonitis. Peritonitis was induced by intraperitoneal injection of 500 μL 4% Brewer thioglycollate medium (BD Difco). Seventy-two hours after challenge, the peritoneal cavity was lavaged with 5 mL PBS. Lavage fluid was analyzed by differential cell count and flow cytometry by a blinded investigator as previously described.29 Blood was collected, and plasma fibrinogen levels were quantified as described above.
siFga and experimental metastasis
Female WT mice were injected via tail vein with siFga or siLuc (2 mg/kg) 21, 14, 7, and 0 days prior to tumor cell inoculation at Cincinnati Children's Hospital Medical Center. Thirty minutes after the last siRNA-LNP injection, mice were injected with 300 μL of GFP-expressing Lewis lung carcinoma (LLCGFP) cells (3.0 × 105 cells) via tail vein and then euthanized 14 days later. The cells were grown in complete medium (Dulbecco’s modified Eagle medium, 10% FBS, 2 mM L-Glut, 2% Pen/Strep) for at least 1 passage and reached 70% confluency. The cells were harvested by brief exposure to trypsin/EDTA, washed, and resuspended in ice-cold PBS. Fourteen days after tumor inoculation, lungs were harvested, and pulmonary LLCGFP foci were counted by a blinded investigator using a fluorescent microscope. Blood was collected from a subset of mice 14 days after LPS challenge, and plasma fibrinogen levels were determined as described above.
Statistical analyses
A Shapiro-Wilkes test was performed to determine whether data were normally distributed. Pairwise comparisons were performed with 2-tailed unpaired Student t test or Mann-Whitney U test. Comparisons between multiple groups with 1 variable were performed by regular 1-way analysis of variance (ANOVA) followed by Tukey’s multiple comparison test for normally distributed and unpaired data. Data not normally distributed were compared by Kruskal-Wallis (unpaired) or Friedman (paired) test, followed by Dunn’s multiple comparison tests. Two-way ANOVA followed by Tukey’s multiple comparison test was used to compare data sets with 2 variables. All statistical comparisons were performed with Graphpad Prism 9 (Graphpad Software). For cytokine data sets, values that were out of range of the standard curves were treated as partially observed values (censored observation approach) using R to obtain imputed values.30
Results
LNP delivery of siFga controllably depletes plasma fibrinogen in mice and knocks down human FGA mRNA in vitro
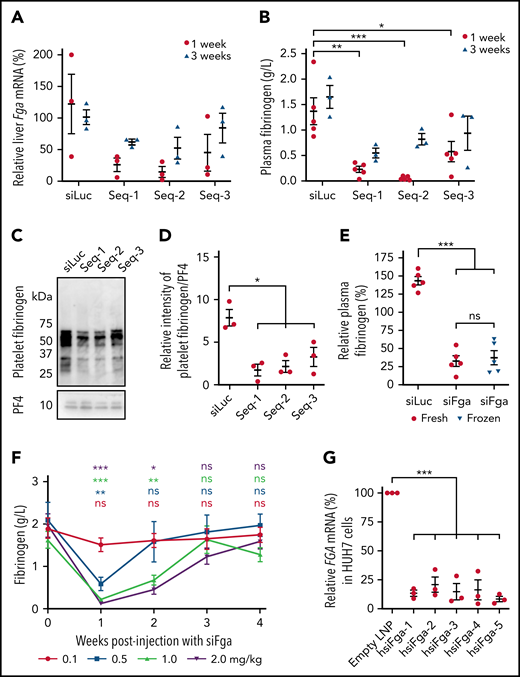
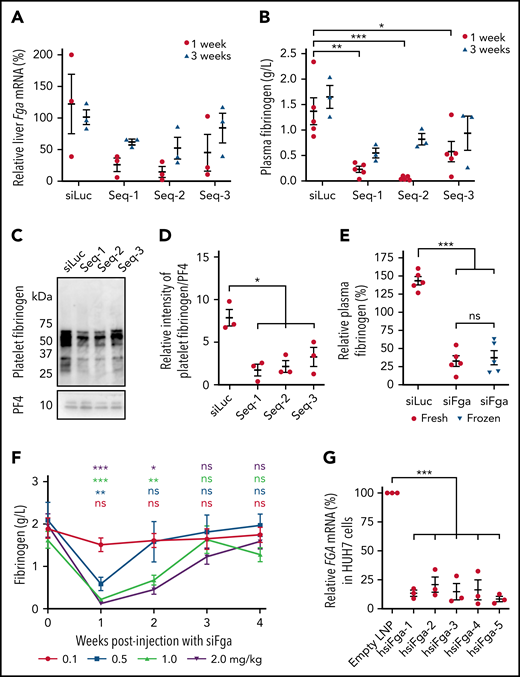
Three siRNA sequences targeting different regions of murine Fga mRNA were designed in silico (Seq-1, Seq-2, and Seq-3), encapsulated in LNPs, and administered to mice at a dose of 1 mg/kg. One week after injection, significant knockdown of hepatic Fga mRNA was observed for each sequence (Figure 1A). This was associated with a significant decrease in plasma fibrinogen levels for all 3 sequences compared with mice treated with siLuc (Seq-1 = 0.22 0.06 g/L, Seq-2 = 0.05 0.02 g/L, Seq-3 = 0.57 0.20 g/L, and siLuc = 1.37 0.26 g/L) (Figure 1B). Decreased fibrinogen levels were still evident 3 weeks after injection, although both hepatic Fga mRNA levels and plasma fibrinogen began to recover at this time point (Figure 1A-B). Fibrinogen levels in platelets were also significantly decreased 1 week after administering the siRNAs (Figure 1C-D). The most potent sequence, Seq-2 (hereafter referred to as siFga), was used in all subsequent studies. We also developed a formulation of siFga that could be frozen for long-term storage; this was equally effective at depleting plasma fibrinogen as the fresh formulation (Figure 1E).
siFga controllably decreases plasma concentration of fibrinogen in mice and knocks down human FGA mRNA in vitro. (A-E) Mice were injected with a single dose of siRNA targeting mouse Fga mRNA (Seq-1, Seq-2, and Seq-3) or siLuc as control. (A) Hepatic Fga mRNA levels 1 and 3 weeks postinjection, normalized to a housekeeping gene (Ppia) and graphed relative to Fga mRNA levels in siLuc-treated mice (n = 3). (B) Seq-1-3 all significantly knocked down plasma fibrinogen, measured by ELISA (n = 5; 1 week) and (n = 3; 3 weeks). (C) Representative western blots of fibrinogen and PF4 in platelets from mice 1-week postinjection with siLuc, Seq-1, Seq-2, or Seq-3. (D) Quantifying densitometry of bands in panel C (n = 3). (E) Mice were treated with fresh (red) or frozen (blue) formulation of siFga and plasma fibrinogen was quantified 1 week later by ELISA (n = 5). (F) Mice were treated with 0.1 (red), 0.5 (blue), 1.0 (green), or 2.0 (purple) mg/kg of siFga, and blood was collected retro-orbitally weekly post injection to quantify fibrinogen levels by ELISA. Plasma fibrinogen at each week was compared with blood collected prior to siFga injection (week 0; n = 5). (G) HUH7 cells were transfected with human-specific siFga (hsiFga-1, -2, -3, -4, or -5), or empty LNPs as negative control. FGA mRNA was quantified a day later, normalized to a housekeeping gene (Act; n = 3). *P < .05, **P < .01, ***P < .001. ns, no significant difference. Error bars represent mean ± standard error of the mean (SEM).
siFga controllably decreases plasma concentration of fibrinogen in mice and knocks down human FGA mRNA in vitro. (A-E) Mice were injected with a single dose of siRNA targeting mouse Fga mRNA (Seq-1, Seq-2, and Seq-3) or siLuc as control. (A) Hepatic Fga mRNA levels 1 and 3 weeks postinjection, normalized to a housekeeping gene (Ppia) and graphed relative to Fga mRNA levels in siLuc-treated mice (n = 3). (B) Seq-1-3 all significantly knocked down plasma fibrinogen, measured by ELISA (n = 5; 1 week) and (n = 3; 3 weeks). (C) Representative western blots of fibrinogen and PF4 in platelets from mice 1-week postinjection with siLuc, Seq-1, Seq-2, or Seq-3. (D) Quantifying densitometry of bands in panel C (n = 3). (E) Mice were treated with fresh (red) or frozen (blue) formulation of siFga and plasma fibrinogen was quantified 1 week later by ELISA (n = 5). (F) Mice were treated with 0.1 (red), 0.5 (blue), 1.0 (green), or 2.0 (purple) mg/kg of siFga, and blood was collected retro-orbitally weekly post injection to quantify fibrinogen levels by ELISA. Plasma fibrinogen at each week was compared with blood collected prior to siFga injection (week 0; n = 5). (G) HUH7 cells were transfected with human-specific siFga (hsiFga-1, -2, -3, -4, or -5), or empty LNPs as negative control. FGA mRNA was quantified a day later, normalized to a housekeeping gene (Act; n = 3). *P < .05, **P < .01, ***P < .001. ns, no significant difference. Error bars represent mean ± standard error of the mean (SEM).
Next, we evaluated the impact of both dose and time on fibrinogen knockdown after siFga treatment. The lowest dose tested (0.1 mg/kg siFga) had no significant effect on plasma fibrinogen levels (Figure 1F). In contrast, mice treated with 0.5, 1.0, or 2.0 mg/kg siFga had a dose-dependent decrease in plasma fibrinogen levels 1 week after administering siFga (32%, 14%, and 5% of baseline, respectively). Plasma fibrinogen levels returned to baseline levels by the fourth week in all groups, with the highest dose showing the slowest recovery of plasma fibrinogen levels.
Last, we tested whether human-specific siFga can knock down FGA mRNA in vitro in a human hepatoma cell line, HUH7, a well-established experimental substitute for human primary hepatocytes. FGA mRNA was significantly knocked down in cells transfected with LNP containing each of the 5 tested hsiFga, compared with cells treated with empty LNPs (Figure 1G).
Transient fibrinogen knockdown impairs hemostasis with minimal bleeding risk
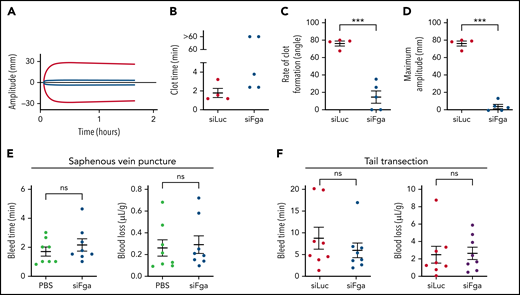
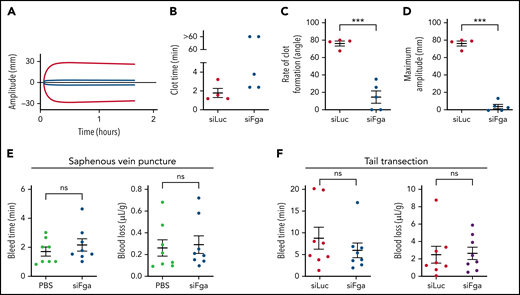
To investigate whether transient knockdown of fibrinogen impairs hemostasis, TEG was performed on whole blood collected from mice treated with 1 mg/kg siFga or siLuc 1 week prior. Both the rate of clot formation and the clot strength were significantly decreased in whole blood from siFga-treated mice (Figure 2A-D). To determine if fibrinogen knockdown was associated with increased bleeding risk in vivo, we compared bleeding times and blood loss following saphenous vein puncture and tail transection in mice treated with 1 mg/kg siFga or control (PBS or siLuc) 1 week prior. Both the bleeding time and the amount of blood loss did not differ between siFga-treated or control-treated mice, in both models (Figure 2E-F).
siFga impairs clot stability ex vivo but does not increase bleeding risk. (A) Representative TEG curve comparing clotting of whole blood from mice treated with siFga (blue) or siLuc (red) 1 week prior. (B-D) Quantifying TEG data such as from panel A to determine clot time (B), rate of clot formation (C), and maximum clot amplitude (D) (n = 4-5). (E) Mice were injected with PBS (green) or siFga (blue) 1 week prior to saphenous vein puncture (n = 8). Bleeding from the saphenous vein injury was monitored for 40 minutes, and bleed time and blood loss were quantified. (F) Mice were injected with siLuc (red) or siFga (blue) 1 week prior to tail transection. Bleeding was monitored for 20 minutes, and bleed time and blood loss were quantified (n = 8). ***P < .001. ns, no significant difference. Error bars represent mean ± SEM.
siFga impairs clot stability ex vivo but does not increase bleeding risk. (A) Representative TEG curve comparing clotting of whole blood from mice treated with siFga (blue) or siLuc (red) 1 week prior. (B-D) Quantifying TEG data such as from panel A to determine clot time (B), rate of clot formation (C), and maximum clot amplitude (D) (n = 4-5). (E) Mice were injected with PBS (green) or siFga (blue) 1 week prior to saphenous vein puncture (n = 8). Bleeding from the saphenous vein injury was monitored for 40 minutes, and bleed time and blood loss were quantified. (F) Mice were injected with siLuc (red) or siFga (blue) 1 week prior to tail transection. Bleeding was monitored for 20 minutes, and bleed time and blood loss were quantified (n = 8). ***P < .001. ns, no significant difference. Error bars represent mean ± SEM.
LPS-induced hyperfibrinogenemia is attenuated with siFga
Mice injected with LPS develop increased levels of plasma fibrinogen, and markers of activated coagulation and inflammation.31,32 To determine if fibrinogen knockdown would attenuate proinflammatory responses following an acute inflammatory challenge, mice were treated with 1 mg/kg siLuc or siFga and then challenged with either 10 mg/kg LPS, or PBS, 1 week later. Hepatic Fga mRNA levels were significantly decreased in siFga-treated mice compared with siLuc-treated mice 24 hours after LPS challenge, whereas hepatic Fgb mRNA levels were significantly increased in both groups compared with their respective PBS-treated cohorts (Figure 3A-B). Pretreatment with siFga significantly attenuated the increase in Fgg mRNA after LPS challenge but did not knock down Fgg mRNA in the PBS control groups (Figure 3C). siFga pretreatment resulted in a significant decrease in plasma fibrinogen, from 3.78 0.17 g/L in the siLuc control group to 1.00 0.21 g/L with siFga after LPS challenge (Figure 3D). D-dimer levels increased after LPS challenge with siLuc, but not with siFga (Figure 3E). The plasma levels of IL-1β, IL-17, TNF-α, MCP-1, and MIP-1α were also quantified (Figure 3F). Compared with the respective siLuc pretreated groups, mice pretreated with siFga had lower levels of each cytokine after treatment with PBS, and except for IL-1β after treatment with LPS, although the differences were not statistically significant for any particular cytokine.
siFga attenuates acute phase response following LPS challenge. Mice were treated with siLuc (red) or siFga (blue) 1 week prior to intraperitoneal injection with LPS and euthanized 24 hours later. (A-C) Relative liver Fga (A), Fgb (B), and Fgg (C) mRNA levels in mouse livers, normalized to a housekeeping gene (B2m). (D-E) Plasma fibrinogen (D) and D-dimer (E) levels were quantified by ELISA. (F) TNF-α, IL-1β, IL-17, MCP-1, and MIP-1α levels in plasma were quantified using a multiplex cytokine analysis. n = 9 for panels A, B, and D; n = 6 for panels C and F, and n = 3 for panel E. *P < .05, **P < .01, ***P < .001. ns, no significant difference. Error bars represent mean ± SEM.
siFga attenuates acute phase response following LPS challenge. Mice were treated with siLuc (red) or siFga (blue) 1 week prior to intraperitoneal injection with LPS and euthanized 24 hours later. (A-C) Relative liver Fga (A), Fgb (B), and Fgg (C) mRNA levels in mouse livers, normalized to a housekeeping gene (B2m). (D-E) Plasma fibrinogen (D) and D-dimer (E) levels were quantified by ELISA. (F) TNF-α, IL-1β, IL-17, MCP-1, and MIP-1α levels in plasma were quantified using a multiplex cytokine analysis. n = 9 for panels A, B, and D; n = 6 for panels C and F, and n = 3 for panel E. *P < .05, **P < .01, ***P < .001. ns, no significant difference. Error bars represent mean ± SEM.
Controlled fibrinogen knockdown restores macrophage migration in plasminogen-deficient mice
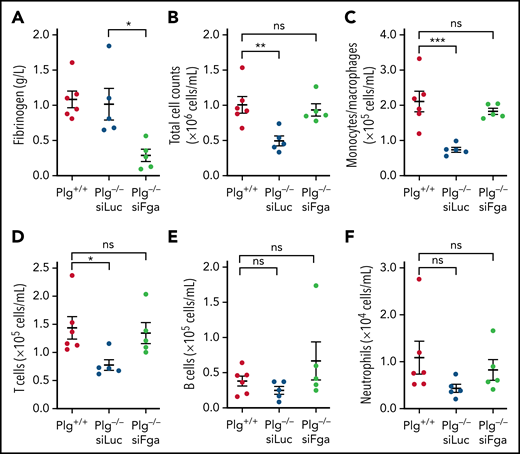
Plg−/− mice have impaired leukocyte migration that is restored by fibrinogen deficiency, or by mutating the integrin αMβ2-binding site on fibrinogen to prevent fibrinogen-leukocyte interaction.29 We tested if controlled fibrinogen knockdown could also restore impaired macrophage migration in a model of peritonitis. Plg−/− mice were pretreated with 1 mg/kg siLuc or siFga 1 week prior to a thioglycollate injection into the peritoneal cavity. Fibrinogen was significantly depleted in Plg−/− mice pretreated with siFga (Figure 4A). As expected, the peritoneal lavage fluids from Plg−/− mice pretreated with siLuc had significantly fewer total leukocytes compared with Plg+/+ after stimulating with thioglycollate (Figure 4B). This was predominantly due to impaired monocyte/macrophage migration to the peritoneal cavity (0.73 0.07 × 105 per mL of peritoneal lavage fluids) compared with Plg+/+ mice (2.11 0.29 × 105 cells per mL) (Figure 4C). Pretreatment with siFga restored monocyte/macrophage migration into the peritoneal cavity (1.83 0.08 × 105 cells per mL) to levels similar to Plg+/+ mice. Plg−/− mice pretreated with siLuc also had slightly less T cells recruited to the peritoneal cavity compared with Plg+/+ mice, which was also restored by pretreatment with siFga (Figure 4D). Levels of B cells and neutrophils did not differ between Plg+/+ and siLuc pretreated Plg−/− mice (Figure 4E-F).
siFga restores macrophage migration in plasminogen-deficient mice. Plasminogen-deficient (Plg−/−) mice were injected with siLuc (blue) or siFga (green) 1 week prior to intraperitoneal injection of thioglycollate and euthanized 72 hours later. Plasminogen WT littermates (Plg+/+; red) were included as control and were also injected with thioglycollate. (A) Plasma fibrinogen levels were quantified by ELISA. (B-F) Peritoneal lavage fluids were analyzed by flow cytometry for total cell counts (B), monocyte/macrophages (C), T cells (D), B cells (E), and neutrophils (F). n = 5 to 6 all panels. *P < .05, **P < .01, ***P < .001. ns, no significant difference. Error bars represent mean ± SEM.
siFga restores macrophage migration in plasminogen-deficient mice. Plasminogen-deficient (Plg−/−) mice were injected with siLuc (blue) or siFga (green) 1 week prior to intraperitoneal injection of thioglycollate and euthanized 72 hours later. Plasminogen WT littermates (Plg+/+; red) were included as control and were also injected with thioglycollate. (A) Plasma fibrinogen levels were quantified by ELISA. (B-F) Peritoneal lavage fluids were analyzed by flow cytometry for total cell counts (B), monocyte/macrophages (C), T cells (D), B cells (E), and neutrophils (F). n = 5 to 6 all panels. *P < .05, **P < .01, ***P < .001. ns, no significant difference. Error bars represent mean ± SEM.
Transient fibrinogen knockdown inhibits the metastatic potential of circulating tumor cells
Fibrin(ogen) is an important contributor to the metastatic potential of circulating tumor cells.7,8
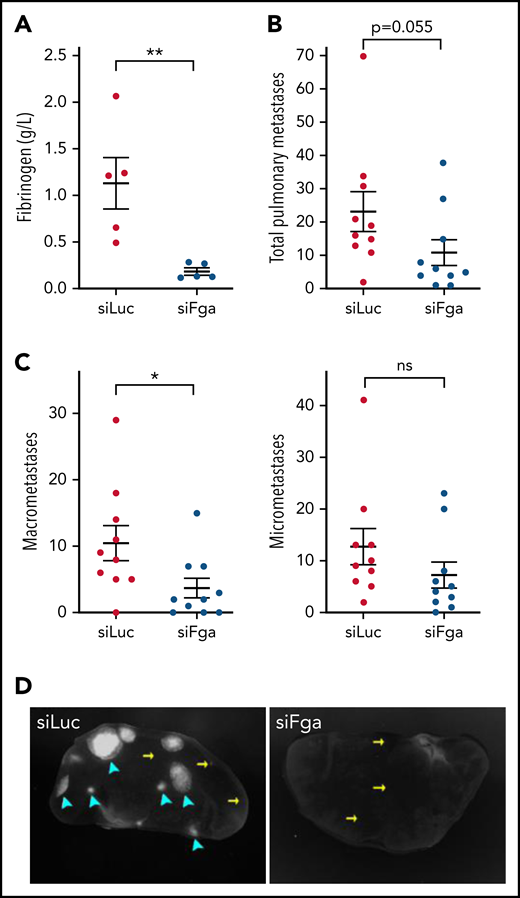
Using a mouse model of experimental metastasis, we determined whether transiently decreasing plasma fibrinogen levels with siFga affects the metastatic potential of circulating tumor cells. To significantly deplete plasma fibrinogen before inoculating the mice with tumor cells, mice were treated with 2 mg/kg siLuc or siFga weekly starting 3 weeks prior to tumor cell injection. Two weeks after tumor cell injection, mice pretreated with siFga had significantly lower plasma fibrinogen levels (Figure 5A) and decreased number of pulmonary metastases (10.9 4 metastases) compared with siLuc-treated animals (23.2 6 metastases) (Figure 5B). This reduction was mostly driven by a significant decrease in macrometastases in the lungs of siFga-treated mice (Figure 5C-D). The number of micrometastases in the lungs appeared to decrease in siFga-treated mice, although not significantly at the sample size tested.
siFga decreases the formation of pulmonary macrometastases in a model of experimental metastasis. Mice were injected with siLuc (red) or siFga (blue) weekly for 3 weeks prior to intravenous inoculation with GFP-expressing LLC cells (LLCGFP), and blood and tissue were collected 14 days later. (A) Plasma fibrinogen concentrations were quantified by ELISA (n = 5). (B) Total pulmonary LLCGFP metastases in mice treated with control siLuc or siFga (n = 10). (C) Total number of pulmonary LLCGFP macrometastases (left) and micrometastses (right). (D) Representative images of lung lobes from siLuc- (left) and siFga- (right) treated mice. The cyan arrowheads indicate macrometastases, and the yellow arrows indicate micrometastases. *P < .05, **P < .01. ns, no significant difference. Error bars represent mean ± SEM.
siFga decreases the formation of pulmonary macrometastases in a model of experimental metastasis. Mice were injected with siLuc (red) or siFga (blue) weekly for 3 weeks prior to intravenous inoculation with GFP-expressing LLC cells (LLCGFP), and blood and tissue were collected 14 days later. (A) Plasma fibrinogen concentrations were quantified by ELISA (n = 5). (B) Total pulmonary LLCGFP metastases in mice treated with control siLuc or siFga (n = 10). (C) Total number of pulmonary LLCGFP macrometastases (left) and micrometastses (right). (D) Representative images of lung lobes from siLuc- (left) and siFga- (right) treated mice. The cyan arrowheads indicate macrometastases, and the yellow arrows indicate micrometastases. *P < .05, **P < .01. ns, no significant difference. Error bars represent mean ± SEM.
Discussion
Because fibrinogen is implicated in the pathogenesis of multiple disease processes, strategies for decreasing circulating fibrinogen for long durations may have therapeutic utility. We used an LNP-siRNA strategy to decrease hepatic Fga mRNA expression and confirmed, in multiple cohorts of mice across 5 different institutions, that this results in a significant decrease of plasma fibrinogen by 1 week post-treatment. Intraplatelet fibrinogen was also decreased by 1 week after administering siFga in mice. Mouse platelets have a lifespan of 2 to 5 days,33,34 and this decreased fibrinogen is consistent with receptor-mediated endocytosis of fibrinogen from plasma into platelets.35,36 Varying the siRNA sequence, or the dose of the most potent sequence, resulted in a range of fibrinogen knockdown for 2 to 3 weeks after a single dose. Although not shown here, extending the duration of fibrinogen knockdown can be easily achieved by the well-established approach of repeat dosing of LNP-siRNA at regular intervals. We had previously developed other LNP-siRNA reagents and achieved prolonged (>5 months) knockdown of coagulation factor XIII in mice using this repeat dosing regimen approach.26 Onpattro, the first siRNA therapeutic for the treatment of hereditary transthyretin-mediated amyloidosis in humans, is also administered at regular intervals.25,37
Whole blood collected from mice 1 week after administering 1 mg/kg siFga formed clots with decreased stiffness, as measured by TEG ex vivo. However, this dose did not significantly exacerbate bleeding in either a mouse saphenous vein puncture model or a tail transection model of bleeding. The discrepancy may be because the blood flow through the injured vessels in both models has presumably lower shear force applied to the forming clot compared with TEG. Another potential explanation is compensation by other plasma or platelet proteins, such as fibronectin.38,39Fga knockout mice have undetectable plasma fibrinogen, but fatal spontaneous bleeding events are rare in adult mice.38 Fibronectin is vital to the control of bleeding with fibrinogen deficiency, supported by increased blood loss from mice with dual knockout of fibrinogen and fibronectin in a tail transection model compared with fibrinogen deficient mice.40 It is also possible that the residual plasma fibrinogen after siFga treatment was sufficient to maintain hemostasis in both bleeding models at the dose tested. Fibrinogen as low as 0.5 to 1 g/L appears to be adequate for hemostasis in humans.41 In fibrinogen-deficient patients with unprovoked bleeds, replacement therapy is initiated and maintained so that fibrinogen levels are >1 g/L until bleeding stops, and >0.5 g/L until the wound is healed, whereas secondary prophylaxis to maintain a trough level of >0.5 g/L is crucial in patients with a personal or family history of bleeds, or severely deficient factor levels.42,43 Furthermore, 1 g/L fibrinogen is often recommended as a threshold for initiating fibrinogen replacement therapy to treat active bleeding or as a target level to achieve prior to surgery, although higher target fibrinogen levels (1.5-2.0 g/L) are recommended for management of patients with life-threatening hemorrhage.44-46 Additional experiments, such as in larger animals and in severe bleeding models, are needed to better understand the bleeding risk when fibrinogen is knocked down to human-equivalent levels of 0.5 to 1 g/L by siFga.
Fibrinogen is an acute phase reactant that also contributes to host inflammatory responses by affecting leukocyte biology and trafficking, and by stimulating the secretion of proinflammatory cytokines.2,4,47-49 In a standard model of inflammation induced by LPS, pretreatment with siFga prevented upregulation of hepatic Fga mRNA, leading to decreased levels of plasma fibrinogen and D-dimer 24 hours following LPS challenge. Mice treated with siFga also had lower levels of TNF-α, IL-17, MCP-1, and MIP-1α, although the differences were not statistically significant for any particular cytokine. We measured cytokine levels 24 hours after the LPS challenge, and measuring at earlier times may add further clarity, as the increase in levels of proinflammatory cytokines usually peak at ∼4 hours after challenge with LPS.50 Interestingly, lower cytokine levels were also observed in fibrinogen-depleted mice even in the absence of inflammatory challenge with LPS. These results warrant further investigation but suggest that the levels of proinflammatory cytokines in other pathological conditions could potentially be attenuated by depletion of plasma fibrinogen with siFga. For example, COVID-19–associated coagulopathy is associated with dramatic increase in fibrinogen of 5 to >10 g/L, significant inflammation, and high incidence of thrombosis in severely ill patients.9,10,51 Thromboprophylaxis or treatment with thrombolytics has been challenging for patients with COVID-19 because of risks of bleeding and only transient improvement in symptoms.52-54 Furthermore, incidence of thrombosis remains high despite thromboprophylaxis,55 highlighting the need for additional approaches to alleviate thromboinflammation. Given that fibrinogen is significantly upregulated during inflammation,1,2,4 and that hyperfibrinogenemia increases the risk for thrombosis,10,17 depletion of circulating fibrinogen back to normal levels could be an alternate therapeutic strategy to manage thromboinflammation in many diseases, including in COVID-19.
Fibrin(ogen) can directly bind to cell surface receptors on leukocytes, such as macrophages. Plasmin-mediated fibrinolysis disrupts this interaction and enables macrophages to migrate to sites of inflammation.4,29,48,49Plg−/− mice have impaired macrophage migration, but this phenotype is rescued by fibrinogen deficiency, or preventing fibrin(ogen)-leukocyte interaction by mutating the leukocyte integrin binding motif on fibrinogen.29 We demonstrated that decreasing plasma fibrinogen using siFga was sufficient to restore macrophage migration in Plg−/− mice following thioglycollate-induced peritonitis. In addition to establishing the efficacy of siFga in modulating another fibrinogen-dependent disease process, this work also highlights the utility of siFga as a research tool. Genetic fibrinogen knockout mouse models are available, but obtaining a cohort for experiments may be difficult because of different institutional regulations, and maintaining a breeding colony is a time-consuming process. Imposing fibrinogen deficiency in other genetically modified mice requires extensive breeding, is costly and laborious, and may not be feasible if the resulting double or triple knockouts are not viable or fertile. Our LNP-siRNA strategy has clear benefits over the traditional genetic knockout models. A simple IV injection of LNPs containing siFga effectively results in fibrinogen deficiency within 1 week in both WT and transgenic mice. The extent and duration of knockdown can be easily controlled by tailoring the dosing regimen, which has major implications for determining the threshold levels of fibrin(ogen) in the target disease models. This approach can also be easily adapted for depleting fibrinogen in other species, by identifying optimal siFga sequences targeting the species-specific Fga mRNA; we successfully knocked down FGA mRNA in human cells in vitro using this LNP-siRNA strategy.
Fibrin(ogen) supports metastasis by blocking the ability of NK cells to clear tumor cells. Consistent with this, fibrinogen-deficient mice form less pulmonary foci in a model of experimental metastasis.7,8 Using a similar model, we demonstrated that controlled depletion of plasma fibrinogen using siFga prior to inoculating mice with tumor cells decreased the formation of pulmonary metastases. This finding highlights another potential application for siFga; surgery is a common and important intervention for many patients with cancer but is associated with an increased risk of metastasis.56 Administering siFga to controllably decrease fibrinogen in the preoperative period is a potential strategy to improve long-term outcome in patients with cancer by minimizing metastasis in the perioperative period.
In summary, siFga was highly effective at decreasing the plasma concentration of fibrinogen for up to 2 to 3 weeks after a single bolus dose. Decreasing fibrinogen is expected to be safe if kept above a concentration of 1 g/L, the target threshold for initiating fibrinogen replacement therapy. As we have shown, the dosing regimen of siFga can be tailored to achieve tunable knockdown and ensure fibrinogen remains above this target level. The work presented here demonstrates broad applicability of this reagent both as a powerful experimental tool to investigate the role of fibrin(ogen) in (patho)physiological processes and as a potential therapeutic to manage fibrin(ogen)-dependent pathologies, although further work is needed to establish its clinical utility.
Acknowledgments
The authors thank Abdohllah Safari for help with statistical analyses for Figure 3F.
This work was funded by the Canadian Institutes of Health Research grants FDN-148370 and MSH-130166 (C.J.K.), the Michael Smith Foundation for Health Research grant 16498 (C.J.K.), the Canadian Foundation for Innovation grant 31928 (C.J.K.), the BC Knowledge Development Fund (C.J.K.), Nanomedicines Innovation Network of the Networks of Centres of Excellence of Canada grant 2020-T2-02 (C.J.K.), and UBC’s Centre for Blood Research (L.J.J.). This work was also supported in part by US Department of Agriculture, National Institute of Food and Agriculture (J.P.L.). Research reported in this publication was supported by the National Institutes of Health (NIH), through the National Institute of Diabetes and Digestive and Kidney Diseases, under award numbers R01 DK122813 (J.P.L.) and DK112778 (M.J.F.). This work was partially funded by the National Institute of Dental and Craniofacial Research–Intramural Research Program (T.H.B.) and grant 1K99DE030124-01A1 (L.M.S.). Cytokine multiplex analyses were performed by the Advanced Analytics Core (UNC Chapel Hill, NIH grant P30 DK034987).
The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH. The opinions or assertions contained herein are the private views of the authors and are not to be construed as official or as reflecting the views of the Department of the Army or the Department of Defense.
Authorship
Contribution: L.J.J. designed and performed experiments, analyzed and interpreted data, made the figures, and wrote the initial draft of the paper; B.L.P., J.L., and E.M.C. helped perform the experiments; W.S.H., L.M.S., A.W.S., B.F., M.K.R., D.J.G., and J.P.L. designed and performed experiments, analyzed and interpreted data, and edited the paper; A.P.C. provided intellectual input on hypothesis and experimental design and edited the paper; P.R.C., T.H.B., M.J.F., and J.S.P. helped design the experiment, interpreted data, and edited the paper; and C.J.K. designed experiments, interpreted data, and wrote the paper.
Conflict-of-interest disclosure: P.R.C. acknowledges financial interest in Acuitas Therapeutics. C.J.K. and P.R.C. are directors and shareholders of NanoVation Therapeuitcs, Inc, which is developing RNA-based therapies. C.J.K., L.J.J., A.W.S., J.L., M.K.R., and P.R.C. have filed intellectual property on RNA-based therapies, with the intention of commercializing these inventions. A.P.C. is an active duty officer in the US Army and declares no competing financial interests. The remaining authors declare no competing financial interests.
Correspondence: Christian J. Kastrup, 8727 West Watertown Plank Rd, Milwaukee, WI 53226; e-mail: ckastrup@versiti.org.
Requests for data sharing may be submitted to Christian J. Kastrup (ckastrup@versiti.org).
There is a Blood Commentary on this article in this issue.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked "advertisement" in accordance with 18 USC section 1734.