Issue Archive
Table of Contents
BLOOD COMMENTARIES
PERSPECTIVE
Smoldering multiple myeloma: taking the narrow over the wide path?
The optimal treatment approach for patients with smoldering multiple myeloma (SMM) remains undefined. In this Perspective, Avet-Loiseau and Bahlis consider the data from prospective trials to date and conclude that, as yet, we cannot identify patients with SMM at high risk of progression to symptomatic myeloma precisely enough to warrant noncurative therapy. Rather than designing more interventions, the authors argue for more prospective studies of SMM biology, including phenotypic, genomic, and immune environment analyses.
CLINICAL TRIALS AND OBSERVATIONS
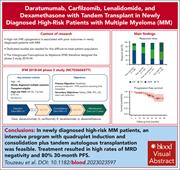
Daratumumab, carfilzomib, lenalidomide, and dexamethasone with tandem transplant for high-risk newly diagnosed myeloma
Clinical Trials & Observations
The optimal management for patients with genetic high-risk multiple myeloma is uncertain, with early relapse being an ongoing challenge. Touzeau and colleagues report the results of the phase 2 Intergroupe Francophone du Myelome 2018-04 trial of quadruplet induction, double autologous stem cell transplantation, and dual-therapy maintenance in these patients. This intensive regimen is feasible and efficacious, achieving a minimal residual disease negativity rate of 94% and a 30-month progression-free survival rate of 80%, and is now being evaluated in a phase 3 randomized trial.
HEMATOPOIESIS AND STEM CELLS
Sickle cell disease iPSC-derived sensory neurons exhibit increased excitability and sensitization to patient plasma
Both acute and chronic pain crises are major challenges for patients with sickle cell disease (SCD) and their pathophysiological basis remains uncertain. Allison et al derived sensory neurons from induced pluripotent stem cells (iPSCs) from patients with SCD and determined that SCD iPSC neurons exhibit multiple indicators of hyperexcitability relative to healthy control iPSC neurons. This work establishes a human-specific model of SCD sensory neurons and suggests that both intrinsic and extrinsic blood-derived factors may underly SCD pain.
LYMPHOID NEOPLASIA
MRD at the end of induction and EFS in T-cell lymphoblastic lymphoma: Children’s Oncology Group trial AALL1231
CME
Clinical Trials & Observations
Brief Report
T-cell lymphoblastic lymphoma is uncommon compared to the related disorder T-cell acute lymphoblastic leukemia, and prognostication is less well established. In this month’s CME article, Hayashi and colleagues analyzed data from a prospective randomized trial in a pediatric, adolescent, and young adult population. The authors found that measurable residual disease (MRD) by flow cytometry of bone marrow at the end of induction treatment was an independent risk factor for event-free survival. MRD warrants evaluation in future studies in this disease where its interaction with tumor genetic features requires exploration.
MYELOID NEOPLASIA
BRG1/BRM inhibitor targets AML stem cells and exerts superior preclinical efficacy combined with BET or menin inhibitor
PLATELETS AND THROMBOPOIESIS
Platelet dysfunction reversal with cold-stored vs room temperature–stored platelet transfusions
Kogler and colleagues report on a randomized crossover study of in vivo platelet function in 10 healthy people taking aspirin after infusion of platelets stored either at room temperature (RT) for 7 days or 4°C for 14 days to evaluate their comparative procoagulant activity. Samples from recipients of cold-stored platelets showed increased thrombin generation but less integrin activation than from those recipients of conventionally stored platelets. The authors also identified assays that predict donors whose platelets retain function during cold storage. Their data reinforce calls for randomized trials of RT- vs cold-stored platelets.
THROMBOSIS AND HEMOSTASIS
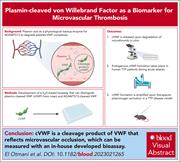
Plasmin-cleaved von Willebrand factor as a biomarker for microvascular thrombosis
Compared to large vessel thrombi, microvascular thrombi are von Willebrand factor (VWF) and platelet enriched but relatively fibrin poor, suggesting incorporation of largely unactivated platelets that do not promote thrombin generation and fibrin deposition. Investigations by El Otmani et al reveal that plasmin plays an important role in degradation of VWF within microvascular thrombi and that a unique plasmin degradation product of VWF may serve as a biomarker of microvascular thrombosis in conditions such as thrombotic thrombocytopenic purpura.
LETTER TO BLOOD
Second primary malignancies after commercial CAR T-cell therapy: analysis of the FDA Adverse Events Reporting System
Clinical Trials & Observations
There are 6 autologous chimeric antigen receptor (CAR) T-cell products currently approved by the US Food and Drug Administration (FDA) for CD19-positive leukemia and lymphoma and BCMA-positive myeloma, and there has been recent concern for second neoplasms occurring in CAR-T recipients. Elsallab et al analyzed the FDA Adverse Event Reporting System and identified 536 second primary malignancies following CAR T-cell therapy, with myelodysplasia, acute myeloid leukemia, and T-cell neoplasms disproportionately common. Additional studies are required to establish the true incidence and causality of these second cancers.
BLOOD WORK
ERRATUM
CONTINUING MEDICAL EDUCATION (CME) QUESTIONS
-
Cover Image
Cover Image
![issue cover]()
Induced pluripotent stem cells from a patient with sickle cell disease (SCD) differentiated into sensory neurons exhibit typical neuronal morphology and expression of the sensory neuron markers peripherin (red), calcitonin gene-related peptide (white), and nuclei (blue). This in vitro model enables the study of pain pathophysiology in SCD. See the article by Allison et al on page 2037.
- PDF Icon Front MatterFront Matter
- PDF Icon Table of ContentsTable of Contents
- PDF Icon Editorial BoardEditorial Board
Advertisement intended for health care professionals
Email alerts
Advertisement intended for health care professionals